
© SSF
Functional feeds could soon form part of the Atlantic salmon sector’s strategy to keep amoebic gill disease at bay. According to a new preliminary trial published in the journal Aquaculture, a nutraceutical aquafeed was able to delay the onset and progression of AGD in salmon smolts. During the feed and disease challenge study, researchers fed AGD-positive and negative control fish a base aquafeed and compared their progression with AGD-positive experimental fish that were fed a customised nutraceutical blend.
Results from the trial showed that the progression of amoebic gill disease was significantly slower in salmon smolts in the experimental group. Fish in the control group displayed more clinical signs of AGD and their infections appeared to be more severe – with lesions covering more of the gill tissue 21 days after infection. The researchers also noted that the nutraceutical blend appeared to stimulate genes that played a role in the salmon’s mucosal defence system. These initial findings suggest that functional feeds could make Atlantic salmon more resistant to AGD infections. It also indicates that enhancing the fishes’ immune function could be part of a new strategy to prevent amoebic infections – potentially saving the salmon industry from costly fish mortalities.
What is amoebic gill disease and how is it treated?
Amoebic gill disease is caused by the opportunistic ectoparasite Paramoeba perurans. The parasite inflames and damages gill tissue in marine fish, leading to breathing difficulties in severe cases, death. Outbreaks often occur on salmon farms and can cause catastrophic economic losses if left untreated.
AGD presents a unique challenge for the salmon aquaculture industry. The two primary treatment options are putting affected salmon into freshwater baths or exposing them to hydrogen peroxide to remove the amoebas. Neither of these treatments work particularly well – gill lesions can reappear within days of therapy. Researchers have also noted that exposing marine fish to freshwater and hydrogen peroxide causes severe osmotic stress, which raises welfare concerns. In addition to these biological concerns, AGD treatments come with a high financial and labour burden for producers.
Developing an effective and ecologically neutral treatment for AGD could revolutionise the industry. As of publication, no vaccines against P. perurans have been created. Some treatments have targeted the parasite, hoping to reduce its ability to colonise gill tissue. Other research projects have tried selectively breeding salmon to make them more resistant to AGD infections. But as re-infection rates remain high and multiple treatments are costly and only partially effective, the industry urgently needs to develop a preventative strategy.
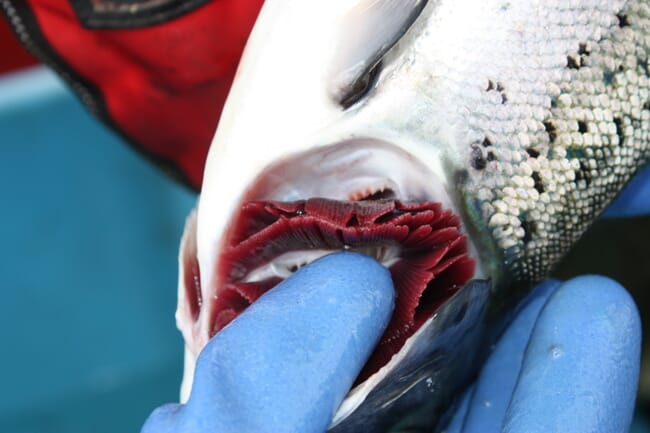
Since there is no vaccine against AGD, researchers exploring other methods to make salmon more resistant to infections. Nutraceuticals are emerging as a promising candidate. These often take the form of supplements in aquafeed that boost the fishes’ immune system function.
Previous studies have shown that nutraceutical strategies have helped Atlantic salmon become more resilient when faced with sea lice infestations. Supplementing aquafeed with immunostimulants has also delayed the onset of diseases like piscine myocarditis and reduced overall viral loads in infected fish.
Testing the nutraceutical strategy

© BioMar
In this study, the research team wanted to investigate whether a new functional fish feed from BioMar could mitigate the severity and progress of AGD infections. The team sourced Atlantic 240 salmon smolts and transferred them to a land-based recirculation facility. The salmon were split into six cohorts – with 40 individuals going into six separate tanks. They were acclimated for seven days before taking part in the 21-day feed and disease challenge trial.
The researchers used a standard feed from BioMar as a control diet (feed A) and as a base feed for their experimental formulation. The researchers supplemented the experimental feed with two micro additive packages to create the nutraceutical formulation (feed B). The fish were feed with 1 percent of their body weight each day.
Fish in four of the six tanks were exposed to 500 amoeba per litre of water for four hours. The two amoeba-free tanks were used as controls to establish a disease-free baseline. The research team took samples of gill tissue from six control fish and six experimental fish after the initial amoeba exposure, after 14 days and at the 21-day mark. The team assessed the presence of amoebic gill disease by using qPCR tests and RNA extraction. qPCR was also used to assess multiple genetic markers and expressions. The team relied on gill examination and scoring to determine the severity of AGD infection. A gill score of 0 (GS0) indicated no AGD signs and a gill score of 5 (GS5) indicated extensive lesions that covered the gill tissue.
The feed and the progression of AGD
The results suggest that nutraceuticals can form a feasible and low-cost preventive strategy for amoebic gill disease. The supplemented feed appeared to slow the infection’s progression in gill tissue. At the end of the trial, the experimental group contained fewer AGD-positive salmon than fish in the control group.
Fourteen days after infection, fish in the control group exhibited notable signs of AGD – the amoeba was detected in qPCR analysis and gill lesions were present. Five of the six fish in this cohort had an AGD gill score of 2 (GS2) – which was considered the humane endpoint for this study. To compare, fish in the experimental group, “exhibited little or no clinical signs of AGD” at the 14-day mark. It took 21 days for salmon on the experimental feed to reach GS2.
Immunostimulants and salmon genetics
When looking closer at the study data, it appears that the experimental feed impacted the expression of genes that regulate the salmon’s mucosal layer and mucosal defence systems. The mucosal layer is a key physical and chemical defensive barrier that protects fish from infections. Stimulating this defence mechanism can prevent pathogens from attaching to gill tissues – a key element in improving resistance to ADG.
Though some “effect” was observed across 10 of the 11 genetic markers the researchers assessed, they couldn’t completely explain why these genes were activated. Nor could they explain the full effects that this activation would have on the salmon’s day-to-day metabolic activities. The relationship between the nutraceutical and mucosal response in the face of an AGD infection needs to be studied before it can be deployed in the field.
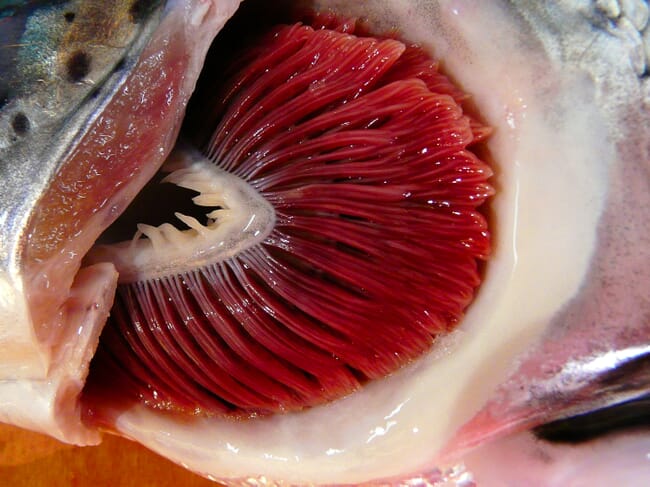
Though these are largely positive results, it should be noted that this was a preliminary trial conducted in a laboratory – not at a working farm site. Studies need to be conducted in an industry setting to validate the findings and to shed further light on the immunity-enhancing effect of the feed.




