Environmental and biological challenges present themselves in every aquaculture enterprise and the culture of Atlantic salmon is no exception, with lice infestation from Caligus species and Lepeophtheirus salmonis, as well as a variety of gill diseases including the proliferative amoebic gill disease (AGD), being the most significant challenges. If not controlled or treated AGD can lead to slow growth, lethargy and often widespread mortality. The focus of this article is to better understand the factors involved in AGD and the relationships between pathogen, host and environment, while the second part will look at the current research status.
History
AGD was first identified as affecting farmed Atlantic salmon in Tasmania in 1984, and has since become a regularly occurring endemic disease there, especially in naïve farmed salmon. So much so that Tasmanian salmon farmers have adapted their production strategies to treat AGD regularly throughout the on-growing phase with the use of freshwater (FW) bathing treatments, administered in wellboats or tarpaulins.
Although hydrogen peroxide (H₂O₂) has been used as a treatment, it is dangerous to fish if used for too long or at high temperatures, causing significant oxidative stress. FW bathing for three to four hours appears much more successful, although access to adequate quantities of FW for wellboat or tarpaulin treatments remains an issue for some sites. Whichever method is used, it will add significant cost to production, normally between 10 and 20 percent.

Ireland first recorded cases of AGD in late autumn 1995, Scotland in 2006 and Norway in late autumn 2006 at four geographically distinct locations. In Norway, histopathological examinations revealed uniform parastomal amoebae in lesions consistent with outbreaks of AGD, although it didn’t cause severe problems until 2013.
Ireland and Scotland’s experiences with the disease have been similar, with severe cases recorded from 2011. Ireland, Scotland, and Norway have all been affected by outbreaks of AGD shortly after introducing both S0 and S1 smolts to sea for on-growing; naïve fish tend to be more susceptible.
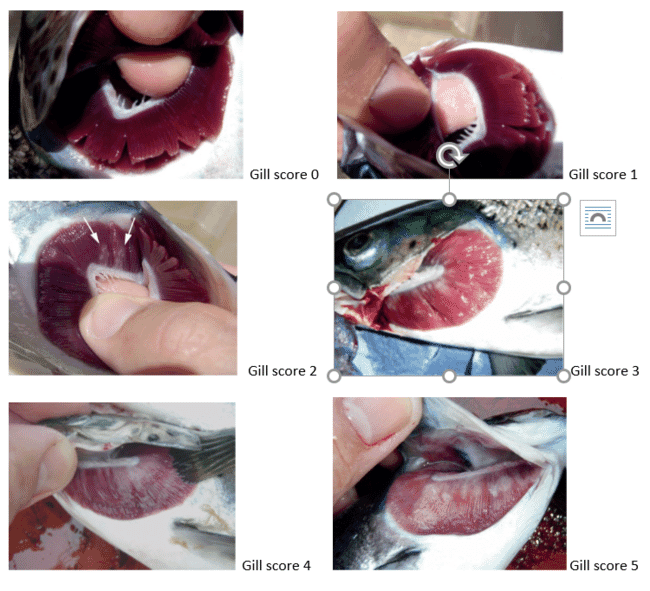
Treatments used to be assigned depending on regular sampling of the gills for gross signs across all eight gill arches (16 hemibranchs) and then compared using a farm-based assessment. However, since 2009, gross signs have been compared visually against a scoring chart as developed and described by Taylor with a score of zero indicating healthy gills and a score of five indicating extensive mucus and lesions covering most of the gill surface (Table 1).
Table 1. Infection, gill score, gross description
Infection level | Gill score | Gross description |
Clear | 0 | No sign of infection and healthy colour |
Very light | 1 | 1 white spot, light scarring or undefined necrotic streaking |
Light | 2 | 2-3 spots/small mucous patch |
Moderate | 3 | Attached thickened mucous patch or spot groupings up to 20% of gill area |
Advanced | 4 | Lesions covering up to 50% of gill area |
Heavy | 5 | Extensive lesions covering most of the gill surface |
When AGD first presented itself in Tasmania in 1984, gross observations, clinical pathology and histopathology (the study of changes in tissues caused by disease) identified two free-living protozoan amoebas, Neoparamoeba pernaquidensis and Neoparamoeba branchiphila as being the likely aetiological agents. However, histopathological gene sequencing using polymerase chain reaction (PCR) assays of non-cultured gill-derived (NCGD) amoeba indicated that neither of these were associated with AGD lesions.
© Mark Adams and Hamish Roger
Instead, a new NCGD amoeba was discovered in 2007, Neoparamoeba perurans, and this soon became known as the likely cause. An amphizoic marine amoeba, N. perurans is a free-living parasite, and will readily attach itself to the gills of salmon and then multiply rapidly by simple cell division. Infection induces hyperplasia and fusion of the lamellar epithelium and formation of interlamellar cysts or vesicles. Histopathology indicates that these lamellae clump together and substantially reduce the gills’ functional surface area, depending on the level of lamellar fusion. This impairment of gill function can lead to hypoxia, acidosis and, ultimately, mortality.
Factors facilitating attachment include changes in the host environment, such as increased salinity and temperature; biological agents such as bacteria, algae, protozoa or jellyfish; and other host factors that may be present and contributing. Therefore, it is thought that there are a number of causes that facilitate the spread of AGD. However, according to Dr Hamish Rodger, global managing director of the Fish Vet Group, while these factors may contribute to AGD, exposing the fish to N. perurans alone without compromise will also give rise to rapid spread of AGD.
Seawater (SW) salinity fluctuations and pH values are intrinsically linked to SW surface temperatures. Although not conclusive, higher SW temperatures are thought to aid the proliferation of N. perurans, leading to AGD outbreaks. Recent evidence suggests that salinity levels greater than 32 parts per thousand (ppt) play an equally important role in the onset of AGD, possibly due to the presence of a multitude of dinoflagellates and diatoms, types of plankton which are associated with waters that have high salinity and which can compromise the gills. If salinities fall below 28 ppt then clinical disease will not appear. AGD proliferates in temperatures of 10°C to 16°C; optimal temperatures for growing Atlantic salmon are 6°C to 16°C. In Norway, the first outbreaks followed SW temperature reaching 3.5°C above normal for that time of year, and in most other outbreaks SW temperatures prior to infection were higher than the seasonal average. However, AGD outbreaks have occurred in Scotland in SW temperatures as low as 7.5°C, indicating that temperature alone is not a causal factor. But higher temperatures will allow AGD to develop much more rapidly and have a more substantial impact.
Recent studies indicate that salmon might develop resistance after first infection. However, from anecdotal experience, if the first treatment is administered when the gill score is at level 1.5 to 2 or above then ongoing infection (and hence treatment) are almost inevitable. In south-west Ireland in 2011-2013, I worked on a site that administered nine FW bathing treatments to the same fish during the on-growing phase, with cumulative mortalities greater than 50 percent).
It could be argued that S1s of 80-100g transferred to sea in April might have an increased chance of resistance to AGD if they were treated in FW baths for up to three hours as soon as gross pathology detects a gill score of 1 to 1.5. Bearing in mind that the main reservoirs for the disease remain farmed salmon, it is imperative that separate year classes are introduced in bays where more than one farm exists, and that appropriate fallowing periods occur to reduce the frequency of treatments.
Early treatment is of utmost importance, before gill scores reach 1 and as soon as histopathological PCR positive results appear. A recent publication by Downes et al indicates that real-time non-destructive molecular diagnostic assays using gill swab samples (as opposed to gill filament samples) can detect the presence of N. perurans within the first 48 hours of infection and thereafter at each sampling. This is an important development in early detection.
Abdon Ryan, husbandry manager for Marine Harvest Ireland South West suggests N. perurans is present in the marine environment all year round and, in agreement with Dr Rodger, thinks seasonal appearances of various micro fauna can compromise the gills and facilitate N. perurans attachment and multiplication. Abdon adds that the peak of production losses in Ireland occurred during the period 2011-2013, with substantial mortality in farms to the north and south. In recent times, mortality due to AGD has reduced substantially – mainly due to a better understanding of the disease and better husbandry procedures. Recently, one site in south-west Ireland introduced S1s in April and as soon as gross pathology indicated gill scores of 1 to 1.5 in June, an FW bath was performed on the whole site, with low mortality. Further treatments were administered in August and October, again with low mortality, and no further treatments were required until harvest. This site experienced 90 percent survival during the on-growing stage due to early and effective treatment. From previous experiences in 2011, if the first treatment is not administered when gill scores are low (less than 1), then subsequent treatments will only serve to lessen the substantial impact of AGD and significant losses will likely occur. Pre-emptive or prophylactic treatments have also been attempted but are of little or no benefit.
A great deal of research has been undertaken on gill diseases and AGD in recent times in attempts to elucidate the cause. This has led to many commentators using the term “complex gill disease” instead of AGD because of the multiple pathogens, including amoebae, that may contribute to gill disease.
Hamish Rodger reports that current research is tending to focus on gill pathogens such as:
a) Epitheliocystis, a condition that affects the gills of many FW and marine fish including salmon, caused by gram-negative bacteria. The main one is thought to be Candidatus Branchiomonas cysticola (Ca. B. cisticola) which has been found and transmitted in both healthy and gill-diseased salmon and is associated with gill pathology.
b) Pox virus has been a major problem in FW fish but has now been detected in marine fish. Salmon gill pox virus (SGPV) may impair innate immunity, facilitating invasion by secondary invaders such as AGD. Apoptosis is an indicator of SGPV being present.
c) A microsporidian parasite of lice and salmon called Desmozoon lepeophtherii may contribute to gill disease but has yet to be confirmed.


