Introduction
Aquaculture is the fastest growing industry producing animal protein, with average annual growth rate of 10 percent during the 1980s and 1990s. For the decade 2012–2022, aquaculture is forecasted to grow between 29 and 50% to fill the growing demand for fish food (FAO 2014). Farming of marine penaeid shrimp boomed from a few 100 MT in the 80s to close to 4 million mt in 2010, with the top five producers being China, Thailand, Vietnam, Indonesia and Ecuador. Despite this apparent success story in terms of production expansion, shrimp production in many regions continues to suffer important economic losses due to the impact of a wide variety of diseases. The white spot syndrome virus (WSSV), one of the main causes of the stagnating shrimp industry in the nineties, continues to affect production in Mexico, Central and South America where the more extensive farming methods in large ponds do not allow effective biosecurity measures against the virus. The shrimp production in SE Asia and Mexico has been ravaged since late 2012 by a new disease known as early mortality syndrome (EMS, or acute hepatopancreatic necrosis disease, AHPND).

Shrimp are actively “grazing” on the bottom substrate, and therefore highly exposed to exchanges of microflora between the environment and the digestive system. This increases the risk for the proliferation of an unfavorable gut microflora, which can affect the optimal functioning of the digestive system. Furthermore, the digestive system of shrimp is the main entry port for bacterial and viral infections, which remain a major risk for the profitability of shrimp production. The use of antibiotics to control microbial developments throughout the production process is not desirable due to the risk for building up resistance and its rejection by legislators and consumers. The shrimp industry requires alternative ways to control the microbial ecosystem in production systems.
Sustainable approaches to modulate the gut microflora in shrimp include the use of a wide variety of natural compounds capable of modulating the microflora towards a favorable composition such as probiotics, organic acids, yeast extracts and phytobiotics.
These strategies may have synergistic effects, for example phytobiotics can enhance the establishment of probiotic bacteria and therefore enhance the efficacy of probiotic inoculations in the production system.
Functional feeds containing gut health promotors deliver with every meal an adequate concentration of natural antimicrobial activities into the shrimp gut. These feeds are a key component of any strategy to prevent diseases, particularly when opportunistic bacteria are a major cause of mortality.
However, the success of this approach will depend on the efficacy of the selected gut health promotor. The gut modulating feed additive ideally is heat stable and can therefore be easily incorporated into the feed at the feedmill and be present in every meal from the starter feed onwards, without requiring major adaptations of the production protocols at the nursery or farm.
Natural feed additives combining different action mechanisms such as direct bactericide/bacteriostatic properties as well as Quorum Sensing inhibition properties at concentrations below MIC, are most promising to reduce the impact from bacterial diseases such as Vibriosis (Coutteau and Goossens, 2014). In the present study, the effect of a feed additive with multiple actions on gut health was evaluated on the productivity in a pilot scale farm in Ecuador.

Conditions and protocol of the farm evaluation
The trial was executed in the experimental station of a shrimp farm located in the Guayas Province, Ecuador during the period February – April 2015. During the trial, two treatments (referred to as “CONTROL” and “SANACORE”) were compared which only differed with regard to the addition of a phytobiotic additive (Sanacore® GM, Nutriad International, Belgium) to the standard feed used at the farm.
The additive was added by top-dressing at 3 g per kg of feed using a commercial binder throughout the trial period. The feed consisted of a commercial feed with 35% protein.
The experimental ponds of approximately 170 m2 were seeded with shrimp of an average size of 70 mg at a density of 10 per m2. The treatment and control were run in 5 and 3 replicated ponds, respectively, during 78 days. The feed was applied once daily during the morning following the same fixed table for all ponds. Pond management followed routine production protocols of the farm.
Production variables at harvest that were compared between treatments included survival, harvested crop yield per ha, feed conversion ratio (FCR), weekly growth and average harvest weight per shrimp.
Production results
Although the inherent variation among ponds did not allow to detect statistically significant differences (P >0.05 for all production variables), the trial showed some interesting trends (Table 1, Fig. 1). The supplementation of the gut health modulator resulted in improved values for survival (with 20.5% compared to control), crop yield (with 14.1%) and food conversion (with 14.9%). The shrimp receiving the feed additive showed 4.7% slower growth.
Table 1: Results at harvest for control ponds and treatment ponds receiving a gut health promotor after 78 days of culture (average and standard deviation of 5 and 3 replicate ponds of 170 m2 for treatment and control, respectively).
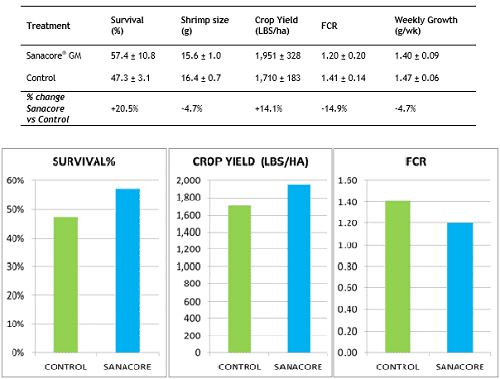
Fig 1: Survival, crop yield and FCR for control ponds and treatment ponds receiving a gut health promotor after 78 days of culture (average and standard deviation of 5 and 3 replicate ponds of 170 m2 for treatment and control, respectively).
Discussion
Natural feed additives combining different action mechanisms against Vibrio species such as direct bactericide/bacteriostatic properties with quorum quenching activities have proven to be effective in improving survival under challenging situations where shrimp are exposed to bacterial pathogens.
The inclusion of such botanical feed additive, the same as the one used in the current study, in a pelletized feed under standard industrial conditions at the feed mill improved survival under production conditions in a semi-intensive shrimp farm in Panama with 24% and 18% compared to the control group during two independent production cycles (Cuellar-Anjel et al., 2011).
In these production trials, the main disease challenge at the farm consisted of WSSV and Vibriosis. The improved survival of 21% relative to control observed in the present study was in line with the observations made in the trials in Panama (Fig. 2). The negative effect on growth observed in the current trials needs further study.
In semi-intensive farming, the use of fixed feeding tables often causes an interference between survival and growth due to the lack of correction of feed availability per shrimp in function of survival. This results often in a growth limitation in the treatment showing the best survival.
Loc et al. (2015) was able to confirm the effect of the same synergistic phytobiotic product in a controlled challenge trial with Vibrio parahaemolyticus (EMS/AHPND strain) under laboratory conditions; showing 62-107% increased survival in shrimp that had received the additive during 3 weeks prior to the experimental infection, compared to unsupplemented control groups. In the latter study by Loc and collaborators, the addition of the phytobiotic product in the diet resulted in consistently lower Vibrio counts in the shrimp’s digestive system compared to the control, illustrating the capability of gut modulating additives to protect the shrimp’s gut flora throughout a Vibrio challenge.
Conclusion
The present study showed at harvest an improvement of survival with 21%, food conversion with 15% and crop yield per ha with 14%, thanks to the use of a functional feed additive capable of modulating the gut flora. This confirmed the potential of functional feeds to improve productivity and reduce costs under the conditions of disease pressures encountered today in most of the production areas in Ecuador, i.e. a combination of WSSV and Vibriosis.
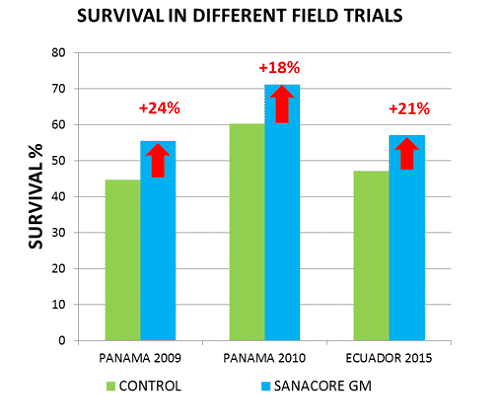
Fig. 2: Comparison of the effect of a functional feed additive with combined anti-bacterial/QS inhibition action on survival of Penaeus vannamei in various field trials under semi-intensive production conditions. Panama: stocking density 8 per m2 in 3ha ponds (Cuellar-Anjel, 2011); Ecuador: present study.
References
- Coutteau, P. and Goossens, T. 2014. Feed additives based on Quorum Sensing Disruption could aid fight against EMS/AHPN. Global Aquaculture Advocate Jan/Feb 2014: p. 84-85.
- Cuéllar-Anjel, J., Vaca, A., Chamorro, R., Dager, S., Coutteau, P. 2011. Effect of a botanical-based feed additive on productivity and economics of semi-intensive shrimp farming in panama. Paper presented at X International Shrimp Culture Symposium & Exhibition. Shrimp Culture Optimization Strategies, “The Returning Path to Success” May 4 – 6, 2011. Panama
- FAO 2014. Economic analysis of supply and demand for food up to 2030 – special focus on fish and fishery products. FAO, Rome.
- Loc H. T., Phuc N.H., Oanh H.B., Trang D.N., Wu M.-H., Ceulemans S., Coutteau P. 2015. Experimental challenge show the potential of functional feed additives for EMS/AHPND prevention. Aqua Culture Asia Pacific Magazine May/June 2015: p. 38-40.
Juan Carlos Valle (1) and Peter Coutteau (2)
(1) Aquaculture Consulting, Guayaquil, Ecuador (jcvalleec@yahoo.com)
(2) Nutriad International, Dendermonde, Belgium (p.coutteau@nutriad.com)




