Seabass and sea bream farming forms the cornerstone of Mediterranean aquaculture. Fry production of these two species has been steadily increasing over the past decades and currently represents more than 90 percent of all fry produced in the Mediterranean. While this has been driven by well-established larviculture and weaning protocols, there is still room for improvement. Developing more efficient protocols that optimise the use of live food creates a window of opportunity that can enhance the quantity and quality of the produced fry while simultaneously reducing operational costs. Moreover, hatchery protocols can also be manipulated to positively modulate the robustness and growth response of future fish. As reported in the following sections, even the smallest breakthroughs in fish larviculture and nursery rearing protocols can optimise performance during grow-out, which should be the ultimate goal of fry production.
Larval rearing protocols
The use of specific products to balance the nutritional and microbiological profile of live feeds is paramount to safeguarding a premium larval performance and maximising stress resistance. A scientific trial was performed with sea bream larvae cultured using an optimised rearing protocol – which included newly developed enrichment for rotifers and Artemia, live-feed conditioners, probiotics and optimised dry-feed protocol. These were compared with sea bream larvae that had been raised with standard commercial products as a control group. While similar survival was recorded between the two sets of larvae at the end of weaning, the biomass achieved using the optimised rearing protocol was significantly higher (Figure 1).
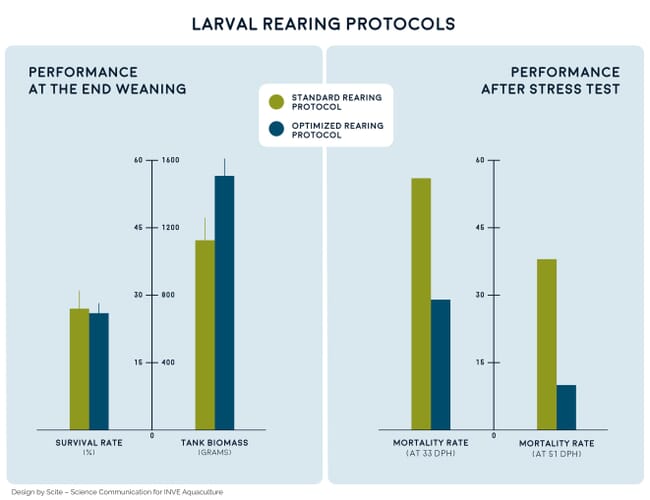
Survival after salinity stress tests was also notably higher when larvae were raised using the optimised rearing protocol (Figure 1). It is also noteworthy that deformity levels were notably reduced in larvae reading using the optimised protocol: 2.3 percent, 1 percent and 4 percent average operculum, tail and severe deformities, respectively, were recorded in the optimised rearing protocol, which contrasts with the 9.3 percent, 6.5 percent and 15.8 percent average deformities, respectively, observed in larvae reared in the control-experimental treatment.
These findings clearly emphasise that one must look beyond traditional end points, such as survival at weaning, to truly understand the impact of improved larviculture protocols. Overall, by optimising larval rearing protocols, one can significantly improve growth, stress resistance and fry quality.
Boosting the immune system through live feeds
For decades, enrichment products were only perceived as a tool to correct nutritional deficiencies of the most commonly used live feeds in marine-fish larviculture, such as rotifers and Artemia. As detailed in the previous instalment, modern hatcheries no longer solely focus on the use of premium enrichment products to fine-tune the nutritional profile of their live feeds. Hatcheries also rely on state-of-the-art microbial-management protocols that boost the quality and performance of their larvae to unprecedented levels.
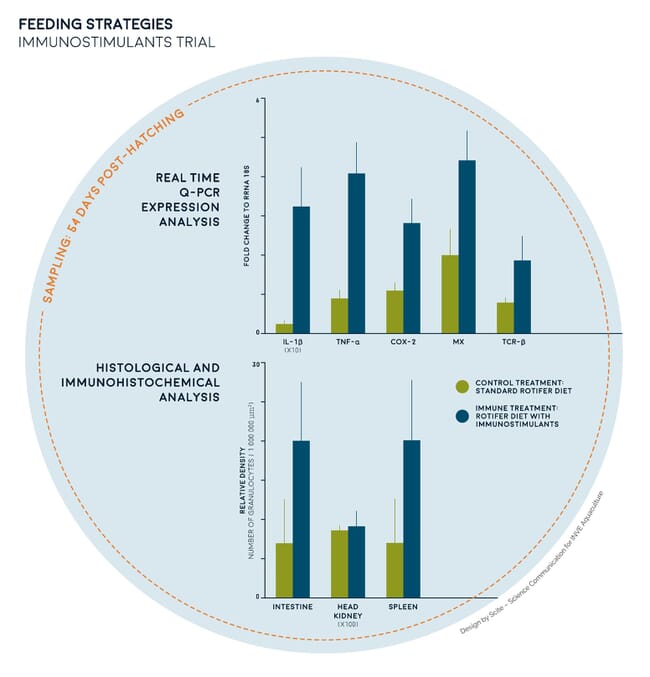
Early-life-specific ingredients are essential to safeguard fry quality. Promoting long-lasting effects during early life can positively affect the physiology and immunology of subsequent life stages, thereby resulting in a superior performance beyond the hatchery gate. This was validated in a scientific trial assessing the effect of manipulating rotifers as a pathway to boost the immune systems of fry. Similar survival and growth were observed for sea bream larvae fed a standard rotifer diet or a diet with rotifers supplemented with specific ingredients. However, larvae that had been fed the supplemented rotifer diet displayed a higher expression level of several immune modulating genes at the end of weaning, with no gut tissue lesions or signs of local inflammation (Figure 2).
These features reveal that specimens provided with this optimised diet are better prepared to respond against stressors, pathogens and diseases. This was confirmed during a salinity stress test at 33 days post-hatching (dph) where sea bream supplied with an optimised rotifer diet displayed a notable reduction in the cumulative stress index (CSI) compared to those fed a standard rotifer diet; while those on the standard diet measured at 124 CSI, those on the optimised diet had just 19 CSI. Overall, results showed that the potential to enhance the immune response of sea bream larvae through fine-tuning rearing protocols and nutritional strategies is only just starting to be understood.
The importance of careful nursery feeds
There is increasing awareness that early-life fish rearing protocols and feed quality can affect performance at later stages. Not only should state-of-the-art larval rearing be combined with genetic improvements, but close attention must also be given to the nursery phase. In fact, sea bream feeding conditions experienced at the nursery stage are decisive for fish performance during cage-based grow-out. Healthy, high quality larvae can suffer if the wrong nursery formulation is used. Feed formulation is paramount to ensure that larvae can grow in a harmonious, healthy and uniform way, rather than purely growing fast. This may only be achieved if optimal feed formulations are employed for each production step: from hatching to post-weaning, nursery and grow-out.
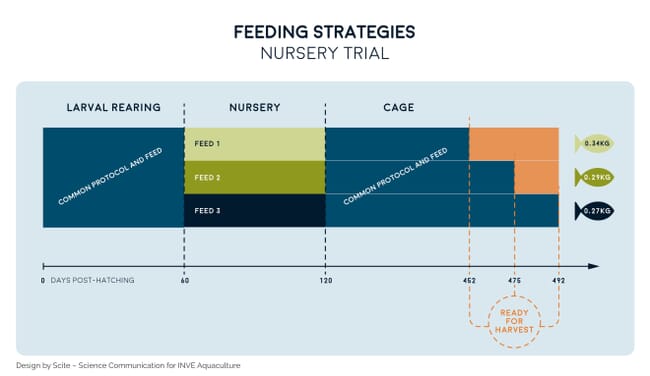
However, formulators making feeds for the post-weaning and nursery stages often erroneously consider the nutritional demand of the fish to be similar to that during grow-out, while in reality nursery stages represent an important step between larval and grow-out stages. This misinterpretation – which endures in marine fish hatcheries – is often the cause of poor performances. Indeed, keeping fish at the nursery stage for an additional five days is a good trade-off if it enables a 50-day gain at grow-out. This was validated in a scientific trial in which sea bream that underwent identical larviculture procedures until 60 dph displayed contrasting grow-out performances in cages if distinct nursery feeds were used (Figure 3).
Nursery feeds supplied during a few weeks (from 60 to 120 days post-hatching) dramatically affect the time that fish take to reach commercial size. Indeed, if a premium nursery feed is used, sea bream can be ready for harvest up to 40 days before those fed on standard grow-out feeds, which force fry to grow without a continuous support of the strategy followed during larval rearing. By safeguarding the supply of an optimal diet at the nursery stage, fish farmers may choose to either harvest earlier or harvest at the standard time point but with a 30 percent weight gain. This weight gain represents a notable reduction in production costs of about 20 percent, which results in a net cost saving of up to €1,000 per tonne of fish produced. It is therefore crucial to support good management during pre-ongrowing to guarantee that the strategy used during larval rearing is continuously supported and that it addresses the fish’s varying demands throughout its life cycle.
Concluding remarks
To foster the farming of seabass and sea bream, and thus leverage Mediterranean and EU aquaculture production towards the goals set for 2030, it is paramount that the industry moves beyond the standardised production protocols currently in use. The partial or even full replacement of live feed, the modulation of the immune response and the promotion of positive carry-over effects from one production stage to the next will play a key role in increasing the productivity and profitability of seabass and sea bream aquaculture.




