Introduction
Importance of Water Quality in Aquaculture
Fish perform all their bodily functions in water. Because fish are totally dependent upon water to breathe, feed and grow, excrete wastes, maintain a salt balance, and reproduce, understanding the physical and chemical qualities of water is critical to successful aquaculture. To a great extent water determines the success or failure of an aquaculture operation.
Physical Characteristics of Water
Water can hold large amounts of heat with a relatively small change in temperature. This heat capacity has far reaching implications. It permits a body of water to act as a buffer against wide fluctuations in temperature. The larger the body of water, the slower the rate of temperature change.
Furthermore, aquatic organisms take on the temperature of their environment and cannot tolerate rapid changes in temperature. Water has very unique density qualities. Most liquids become denser as they become cooler. Water, however, gets denser as it cools until it reaches a temperature of approximately 39ºF. As it cools below this point, it becomes lighter until it freezes (32ºF). As ice develops,water increases in volume by 11 percent. The increase in volume allows ice to float rather than sink, a characteristic that prevents ponds from freezing solid.
Far from being a "universal solvent," as it is sometimes called, water can dissolve more substances than any other liquid. Over 50 percent of the known chemical elements have been found in natural waters, and it is probable that traces of most others can be found in lakes, streams, estuaries, or oceans.
Water Balance in Fish
The elimination of most nitrogen waste products in land animals is performed through the kidneys. In contrast, fish rely heavily on their gills for this function, excreting primarily ammonia. A fish's gills are permeable to water and salts. In the ocean the salinity of water is more concentrated than that of the fish's body fluids. In this environment water is drawn out, but salts tend to diffuse inward. Hence, marine fishes drink large amounts of sea water and excrete small amounts of highly salt-concentrated urine (Figure 1).
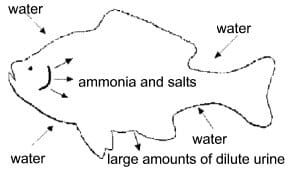
In fresh-water fish, water regulation is the reverse of marine species. Salt is constantly being lost through the gills, and large amounts of water enter through the fish's skin and gills (Figure 2). This is because the salt concentration in a fish (approximately 0.5 percent) is higher than the salt concentration of the water in which it lives. Because the fish's body is contantly struggling to prevent the “diffusion” of water into its body, large amounts of water are excreted by the kidneys. As a result, the salt concentration of the urine is very low. By understanding the need to maintain a water balance in freshwater fish, one can understand why using salt during transport is beneficial to fish.
Figure 1. Direction of water, ammonia, and salt movements into and out of saltwater fish. Saltwater fish drink large amounts of water and excrete small amounts of concentrated urine.
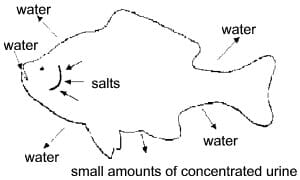
Figure 2. Direction of water, ammonia, and salt movement into and out of freshwater fish. Freshwater fish do not drink water, but excrete large amounts of dilute urine.
Sources of Water
Water is always a limiting factor in commercial fish production. Many of the negative chemical and environmental factors associated with most operations have their origins in the source of water selected. Final site selection has to be made based on both the quality and quantity of water available. The most common sources of water used for aquaculture are wells, springs, rivers and lakes, groundwater, and municipal water. Of the sources mentioned, wells and springs are considered to consistently be of high quality (see AS-486 for more information on water sources).
Water Quantity
The beginning aquaculturist usually underestimates the quantity of water required for commercial production. It is generally accepted that a minimum rate of 13 gallons per minute (gpm) is required for each surface acre of ponds. With this in mind, a 100- acre fish farm will need to have wells capable of producing 1,300 gpm of water. Such large volumes are required to replace water lost to evaporation and seepage. In addition, the farmer may have several ponds to fill quickly during the spawning season. In raceway culture, it is advisable to have a minimum flow rate of 500 gpm. Even water recirculating systems that recycle water require large quantities of water. If a 100,000 gallon capacity water recirculating operation exchanges 10 percent of the water daily, it will require 10,000 gallons of water per day.
The availability of subsurface groundwater in Indiana and Illinois varies widely, ranging from as little as 10 gpm or less to over 2,000 gpm from properly constructed, large diameter wells. With the exception of the aquifers located along major river drainages (usually high yields), potential yields are divided into three distinct regions:
- Northern Indiana and Illinois are good to excellent and, exclusive of some areas near northwestern Indiana, yields from 200-2,000 gpm can be expected.
- In the central portion of Indiana and Illinois, groundwater conditions range from fair to good.
- Well yields from 100-400 gpm are typical for many large-diameter wells. Many areas of southern Indiana and Illinois lack ground-water; generally, less than 10 gpm are available from properly constructed wells. In these areas, the major sources of groundwater are present in the sand and gravel deposits of the river valley aquifers.
These yield potentials do not indicate that an unlimited number of wells can be developed in given location. Detailed studies, including exploratory drilling and test pumping, should be conducted to adequately evaluate the groundwater resource in any given area. The resultant change in the water table is produced by spheres of influence from nearby wells.
Water's Physical Factors
Temperature
After oxygen, water temperature may be the single most important factor affecting the welfare of fish. Fish are cold-blooded organisms and assume approximately the same temperature as their surroundings. The temperature of the water affects the activity, behavior, feeding, growth, and reproduction of all fishes. Metabolic rates in fish double for each 18ºF rise in temperature.
Fish are generally categorized into warmwater, coolwater, and coldwater species based on optimal growth temperatures (Figure 3).
| Figure 3. General temperature ranges for coldwater, coolwater, and warmwater species. | ||
|---|---|---|
| 55-65ºF | 65-75ºF | 75-90ºF |
| Coldwater | Coolwater | Warmwater |
Channel catfish and tilapia are examples of warmwater species. Their temperature range for growth is between 75-90ºF. A temperature of 85ºF for catfish and 87ºF for tilapia is considered optimum.
Walleye, and yellow perch are examples of coolwater species. Ranges for optimum growth fall between 60º and 85ºF. Temperatures in the upper end of this range are considered best for maximum growth for most coolwater species.
Coldwater species include all species of salmon and trout. The most commonly cultured coldwater species in the Midwest is rainbow trout, whose optimal temperature range for growth is 48-65ºF.
Ideally, species selection should be based in part on the temperature of the water supply. Any attempt to match a fish with less than ideal temperatures will involve energy expenditures for heating or cooling. This added expense will subsequently increase production costs.
Temperature also determines the amount of dissolved gases (oxygen, carbon dioxide, nitrogen, etc.) in the water. The cooler the water the more soluble the gas. Temperature plays a major role in the physical process called thermal stratification (Figure 4). As mentioned earlier, water has a high-heat capacity and unique density qualities. Water has its maximum density at 39.2ºF. In spring, water temperatures are nearly equal at all pond depths. As a result, nutrients, dissolved gases, and fish wastes are evenly mixed throughout the pond. As the days become warmer, the surface water becomes warmer and lighter while the cooler-denser water forms a layer underneath. Circulation of the colder bottom water is prevented because of the different densities between the two layers of water. Dissolved oxygen levels decrease in the bottom layer since photosynthesis and contact with the air is reduced. The already low oxygen levels are further reduced through decomposition of waste products, which settle to the pond bottom. Localized dissolved oxygen depletion poses a very real problem to the fish farmer.
Figure 4. Real Media presentation of seasonal changes of water temperatures which occurs in fish ponds. In spring, temperatures and dissolved oxygen are uniform throughout the pond. During the summer, stratification may occur and create an upper layer of water with high-dissolved oxygen and lower layer with low-dissolved oxygen. After a rain or when a phytoplankton die-off occurs the water may turnover.
Pond Turnover Slide Presentation. Please note to view, you will need to have Real Player installed and configured and a sound card with speakers or headphones.
Summer stratification is a greater problem for fish raised in deeper farm ponds. Stratification may last for several weeks. This condition may develop into a major fish kill when sudden summer rains occur. These rains will cool the warmer upper layer of water enough to allow it to mix with the oxygen poor layer below. Decomposing materials in the oxygen-poor layer are again mixed evenly throughout the pond, resulting in an overall reduction in the dissolved oxygen level. Fish previously able to avoid the oxygen depleted layer are now susceptible to low-dissolved oxygen syndrome and possibly death.
Ice is another physical factor directly related to temperature. Normally, ice cover does not impede photosynthesis. Fish consume less oxygen at colder temperatures, greatly reducing the overall oxygen demand. But fish can still suffer from low-dissolved oxygen under snow covered ice. Under extended ice cover, other gases (carbon dioxide, hydrogen sulfide, methane, etc.) can build up to dangerously high levels. Mechanical aeration is probably the most reliable way of preventing an ice buildup by keeping large areas of the pond free of ice.
Suspended Solids
Suspended solids is a term usually associated with plankton, fish wastes, uneaten fish feeds, or clay particles suspended in the water. Suspended solids are large particles which usually settle out of standing water through time. Large clay particles are an exception. Clay particles (which will be discussed again) are kept in suspension because of the negative electrical charges associated with them.
Plankton
Turbidity caused by phytoplankton (microscopic plants) and zooplankton (microscopic animals) is not directly harmful to fish. Phytoplankton (green algae) not only produces oxygen, but also provides a food source for zooplankton and filter feeding fish/shellfish. Phytoplankton also uses ammonia produced by fish as a nutrient source. Zooplankton is a very important food source for fry and fingerlings such as hybrid striped bass and yellow perch. However, excessive amounts of algae can lead to increased rates of respiration during the night thereby consuming extra oxygen. Excessive phytoplankton buildups or "blooms" which subsequently die will also consumes extra oxygen. Any wide swings between day and night oxygen levels can lead to dangerously low oxygen concentrations.
Fish wastes
Suspended fish wastes are a serious concern for water recirculating culture systems. Large amounts of suspended and settleable solids are produced during fish production. As a rule, one pound of fish waste is produced for every pound of fish produced. Fish waste particles can be a major source of poor water quality since they may contain up to 70 percent of the nitrogen load in the system. These wastes not only irritate the fish's gills, but can cause several problems to the biological filter. The particulate waste can clog the biological filter, causing the vitrifying bacteria to die from lack of oxygen. Particulate waste can also promote the growth of bacteria that produces--rather than consumes ammonia.
Clay
Most clay turbidity problems are the result of exposed soil on the pond levee, exposed watershed, crayfish activity, or feeding of bottom dwelling species such as carp and catfish. Turbidity levels exceeding 20,000 ppm can cause behavioral changes in fish. In natural bodies of water, turbidity values seldom exceed these critical levels. Even "muddy looking" ponds rarely have concentrations greater than 2,000 ppm.
Turbidity caused by clay or soil particles, however, can restrict light penetration and limit photosynthesis. Sedimentation of soil particles may also smother fish eggs and destroy beneficial communities of bottom organisms such as bacteria.
Removal of clay turbidity can be accomplished by adding materials that attach to the negative charges of the clay particles, forming particles heavy enough to settle to the bottom. Common remedies for clay turbidity are 7-10 square bales of hay per surface acre, or 300-500 pounds of gypsum per surface acre. Gypsum applications may be repeated at two week intervals if ponds do not clear.
Further Reading
|
|
|
October 2007




