Habitat and biology
The American cupped oyster thrives in estuaries, but also lives in marine coastal environments. It discharges gametes into the water column in response to a variety of stimuli, including warmer temperatures, pheromones, and the presence of appropriate phytoplankton.
Fertilized eggs develop into trochophore larvae that depend on their internal yolk supply for energy. After about 24 hours, the trochophore develops into a shelled veliger larva that uses a ciliated organ (velum) to capture food and to swim.
After two or three weeks, depending on food and temperature conditions, the veliger develops an eyespot and a larval foot (it is now called an eyed larva or pediveliger) with which it explores potential settlement substrates. Upon being stimulated to settle, it cements its left valve to the substrate and metamorphoses into an oyster spat by discarding its velum, reabsorbing its foot, and enlarging its gills.
Production
Production cycle
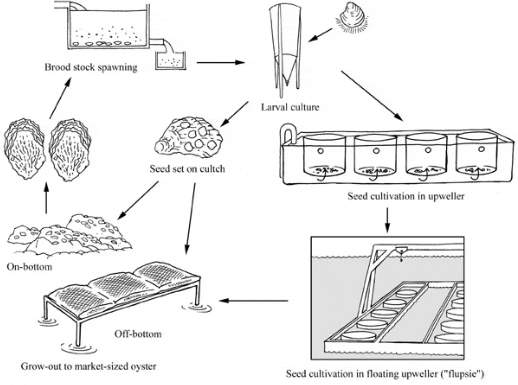
Crassostrea virginica Production Cycle
Production systems
Oyster culture is affected by factors like temperature and salinity, water circulation, the presence and condition of substrate, productivity of appropriate algal food, presence of predators and disease, and protection from ice or storms that might damage culture facilities. Hatcheries allow the culturist to control many of these factors, thus enhancing larval and spat production. Whether oyster cultivation begins in a hatchery or in nature, eventually small oysters are used in either on-bottom or off-bottom (suspended) culture.
Seed supply
North American hatcheries generally use mass culture of algae in tall, clear fibreglass tubes, in very large vats, or in clear, flexible plastic bags from which algae are harvested continuously. The most common cultured algae are the diatoms Chaetoceros calcitrans and Thalassiosira pseudonana (clone 3H Hasle et Heimdal), and a flagellate Isochrysis galbana Tahitian strain (T-iso). Because of the cost of raising enough algae to feed millions of larvae and spat, hatchery-reared oysters are moved into nature as quickly as possible.
Non-ripe adults can be held at low temperatures (usually <20 °C) and fed either natural algae under flow-through conditions or cultured algae in limited flow conditions until they become ready to spawn, usually months before they would in nature. Alternatively, ripe adults can be harvested in nature and brought to the hatchery to be spawned, a process that ends when the spawning season ends in nature.
To initiate spawning, ripe adults are held in containers and heat-shocked with warm water (up to ~30 °C), perhaps with gametes stripped from a ripe adult being added to the containers. Spawning oysters are segregated by sex into smaller containers. After 20 or 30 minutes, the spawned oysters are removed from these containers and the sperm and eggs are washed separately through fine screens that retain debris and oyster tissue expelled during spawning. A small amount of sperm from several males is then mixed with the eggs and fertilization is monitored microscopically to ensure it has been successful. The fertilized eggs are placed in large aerated tanks to develop into trochophore larvae and beyond.
As the larvae grow they are washed every few days through a series of graded sieves that capture different size classes. The larger, faster-growing larvae may be selected; alternatively all larvae may be kept. Those that are kept are placed into new water and fed until they become pediveligers. Subsequently, pediveligers are placed in special setting tanks containing either microcultch (finely ground [~0.025 mm] oyster shell) on which they settle as single cultchless oysters or oyster shell cultch in mesh bags or other containerized systems (these spat usually grow in aggregations). Microcultch systems run water gently downward to keep the larvae within the containers (downwellers) until they settle. Thereafter, the water is reversed to flow upward (upwellers) vigorously through the spat to deliver food, remove wastes, and enhance growth.
Pediveligers may be screened from their culture tanks and held in damp paper or cloth in the refrigerator for a few days in a process called "remote setting". The pediveligers are then shipped in a "cool box" by express transport to culturists who place the larvae in tanks to settle on microcultch or cultch. Thus one hatchery can supply many culturists, none of whom needs to invest in their own hatchery.
Ongrowing techniques
Eventually the culturist must place seed oysters in nature because of the cost of artificially feeding them. Seed oysters, whether produced in hatcheries or harvested in the wild, can be cultured on or off the bottom.
On-bottom culture
As described earlier, the simplest on-bottom culture method does not involve hatcheries. Rather, it depends on transplanting seed oysters to suitable bottom to grow to market size. If the oysters are harvested from public grounds, costs can be low because harvesting fees are usually minimal or non-existent and because the harvest gear is usually inexpensive tongs or dredges. An important expense is the cost of preparing the bottom to receive the seed, perhaps by adding shell or other substrate to stabilize it. Costs increase further if transportation distances are great or if the oysters have to be purchased, either from a commercial harvester or from a hatchery. It may also be costly if environmental conditions inhibit growth, if predation pressure is high, or if mortality from diseases is a problem.
A more complex bottom culture method, bag-and-longline culture, places cultchless seed in mesh bags attached to an anchored line and held intertidally on a hard bottom. Intertidal exposure of the oysters to the air helps protect against predators and fouling organisms that are inhibited or killed by such exposure (fouling organisms compete with the oysters for food and oxygen and add to the weight of the bags, thereby increasing labour or monetary costs of handling the bags).
Off-bottom culture
Bottom culture suffers from the fact that bottom currents tend to be slow, thereby hindering food delivery and waste removal, as well as enhancing sediment deposition onto the oysters, thus interfering with their feeding. Further, many oyster predators live on the bottom. Off-bottom culture ameliorates the negative aspects of bottom culture and makes use of the complete water column, although it is more expensive because it is more labour and technology intensive. Nevertheless, under certain conditions the expense can be justified because of the benefits. For example, in New Brunswick, Canada, with relatively cold ambient water temperatures and a short growing season, a young oyster can reach a commercial size after about 3 years in off-bottom culture compared with up to 8 years in bottom culture.
A number of off-bottom techniques are available, with cultchless spat or seed preferred because cultch is heavy, occupies space, and has high labour and energy costs of handling. Spat may be grown in trays until their shells are strong enough to avoid damage when handled. Bag-and-longline culture holds mesh bags of spat or seed suspended in the water column while attached to an anchored line whereas rack-and-bag culture has the bags fastened to racks that sit above the bottom. Rack culture strings spat-bearing oyster shells on lines, with the shells separated by inert spacers and the lines hanging from racks whereas stake culture impales spat-bearing oyster shells separated by spacers on a stake driven into the substrate. Lantern nets hung from floats are cylindrical containers fashioned from nylon netting and divided into sections in which the oysters are placed. In Stanway tube culture, cultchless oysters in mesh tubes are rotated around an axle by currents.
Harvesting techniques
Cultivation of the American cupped oyster is generally labour-intensive, although mechanical systems are coming into play. For on-bottom culture, oysters can be placed on and harvested by hand from intertidal habitat; hand-operated tongs are used on subtidal beds. Dredges pulled by a vessel are also used on subtidal habitat. Where oysters are grown in bags (on- or off-bottom), in tubes and lantern nets, or on shell-strings, mechanical devices can be used to lift the containers out of the water and onto a vessel for sorting, cleaning, and harvest.
Handling and processing
Oysters harvested for sale are washed or scrubbed clean, then shipped whole to processors. These firms remove (shuck) the soft body (usually done by hand, although mechanical aids are being developed) for added-value processing (e.g. canning, freezing, breading). They may also sell the whole animal to restaurants for the half-shell trade, in which the flatter shell is removed and the soft body is served in the remaining cupped shell (see also Market and trade, below).
Production costs
Recently, it was estimated that production costs for intensive culture (2.5 million seed oysters/hectares) in Florida totalled USD 75 000/ha, with seed, equipment, and labour costing USD 25 000 each. In this situation, oyster survival needs to be 30 percent to achieve break-even if oysters sell for USD 0.10 each.
Diseases and control measures
| DISEASE | AGENT | TYPE | SYNDROME | MEASURES |
|---|---|---|---|---|
| Malpeque Bay Disease | Unknown | Unknown | Mantle regression; gaping; oedema & cream/yellow coloured abscesses up to 1 cm in diameter in body wall; yellow-green scars may also appear on inner surface of shell adjacent to lesions in the soft tissues | Do not introduce oysters from areas which have been affected, historically, by Malpeque disease |
| Dermo Disease; Proliferative Disease; Perkinsosis | Perkinsus marinus | Protozoan parasite | Pale appearance of digestive gland; reduction in condition index; severe emaciation; gaping; shrinkage of mantle away from outer edge of shell; retarded growth; occasionally presence of pus-like pockets; proliferation of parasite causes systemic disruption of connective tissue & epithelial cells; syndrome correlated with warm summer water temperatures (>15-20 °C), when pathogenicity & associated mortalities are highest | No known cure; improved control over transfers; improved management by reducing density & harvesting or moving oysters to low salinity areas (<9‰) before water temperatures increase to 15-20 °C; filter water for hatcheries to 1 µm & treat with ultraviolet light (30 000 µW s ‾¹ cm‾² UV irradiation) |
| Juvenile Oyster Disease (JOD) | Unknown | Unknown | Reduced tissue & shell growth; fragile & uneven shell margins; cupping of the left valve; reduced condition index; mantle retraction & abnormal proteinaceous material (conchiolin) deposits around periphery of mantle on inner shell surface: dying oysters may also display unequal shell growth, with the left valve outgrowing the right valve, & tissue emaciation; can decimate cultured seed | No known preventative measures; can be ameliorated by growing seed past 30 mm as quickly as possible by spawning adults earlier in season, increasing water flow over young spat, & providing food & temperatures that enhance growth; alternatively, spawning adults late in the season also helps; mortality can be reduced or eliminated when oysters are maintained in 25 µm filtered water diluted with high salinity well water; reducing stocking densities within growing trays also improves survival |
| MSX (Multinucleate Sphere X) disease; Haplosporidiosis; Delaware Bay disease | Haplosporidium nelsoni. Its life cycle is unknown | Protozoan parasite | Thin and watery condition, with pale digestive diverticula. A gradual disruption of digestive tubule epithelia may occur, or acute infections may advance so swiftly that there are no clinical signs before death occurs. Mortalities in infected oysters may occur early in the spring, as may infection of new oyster hosts. Mortalities continue through the summer, peaking in August-September. | High mortalities occur rapidly at 18 to 20 ‰, so disease-free oysters should not be introduced to high-salinity habitat. The parasite cannot survive below 10 ‰ salinity, so infections can be rapidly eliminated by placing oysters in mean salinities of ≤10 ‰ and temperatures >20 °C. |
February 2009




