Introduction
The Asian seabass or barramundi, Lates calcarifer (Bloch) is a catadromous fish widely distributed in the tropical Indo-Pacific and an important species for aquaculture (Moore 1982; Cheong 1989; Barlow, Rodgers, Palmer & Longhurst 1993). Vision is the major sense for marine fish larvae to detect zooplankton at sea and in rearing tanks (Blaxter 1986). Vision in fish larvae has been much studied and it is well known that they have pure-cone retinae until metamorphosis, when rod cells develop and the retinomotor response occurs (Blaxter & Jones 1967; Blaxter & Staines 1970; Blaxter 1986; Evans & Browman 2004). Work on vision in Asian seabass has barely started (Yahaya, Lim, Sitti Raehanah, Mukai, Anraku & Kawamura 2011).
It is important to understand the visual sensitivity and behaviour of fish larvae in their natural habitats, and to determine the optimum illuminating conditions in captivity. There are many studies on visual sensitivity of fish larvae through retinomotor responses (Ali 1959; Blaxter 1968a; Blaxter 1969; Blaxter & Staines 1970; Kawamura 1979; Neave 1984; Masuma, Kawamura, Tezuka, Koiso & Namba 2001) and on feeding behaviour under different light intensities (Blaxter 1968a,b; Mukai 2006, 2011; Mukai, Tuzan, Lim & Yahaya 2010). Boeuf and Bail (1999) describe the effect of light intensities on growth of many fish species. Each species requires different light intensities for feeding and predator avoidance, and too intense light is stressful or even lethal. To rear and grow aquaculture fish species like Asian seabass, it is worthwhile to know the required light intensity for different holding conditions, e.g. for larvae in the hatchery. There has yet been no study on the required light intensity for feeding and larval rearing of Asian seabass. Illumination in larval rearing tanks usually has been decided by hatchery technicians, but not based on physiological data such as visual sensitivity and visual thresholds of Asian seabass larvae.
This study examined the retinomotor responses and prey ingestion rates of Asian seabass larvae under various light intensities in the laboratory and determines the visual thresholds and optimum illumination for larval rearing in the hatchery.
Materials and methods
Seabass larvae for experiments
Fertilized eggs of the Asian seabass were obtained from a hatchery of Borneo Marine Research Institute, Universiti Malaysia Sabah. About 10 000 eggs were incubated in a 1 m³ tank with aeration. The eggs hatched within 24 h, and larval rearing was carried out in the same tank at water temperatures 28.9–30.3°C and salinities 25.3–30.2 g L−1. The microalga Nannochloropsis sp. was added daily into the larval rearing tank and maintained at 106 cells mL−1. The larvae were fed rotifers Brachionus sp. and brine shrimp Artemia nauplii. In this article, time was reckoned from hatching, i.e. 1-day-old larvae were sampled next day from hatching, and so on.
Illumination system
The three experiments described in the following sections were all performed in a dark-room laboratory with an illumination system that allowed exposure of larvae to eight light intensities of 0, 0.001, 0.01, 0.1, 1, 10, 100 and 1000 lx. Light was provided by 20-w fluorescent lamps (Power-Glo; Hagen, Montreal, Canada). Light intensity was adjusted by means of neutral density filters (HOYA, NDx8). Light intensities were measured using a light metre (No. 401036; Extech Instruments Co, Nashua, NH, USA) .
Determination of retinomotor response by histology
Asian seabass larvae 10, 15 and 20 days of age were tested for retinomotor response under eight light intensities (0–1000 lx) and processed for retinal examination (total n = 72 larvae). For each age group (n = 24), three larvae were placed in 2-L glass basin and exposed for 90 min under each light intensity.
The larvae were then anaesthetized in 200 mg L−1 MS-222 (ethyl 3-aminobenzoate, methanesulfonic acid salt) and fixed in Bouin's solution under the test light intensity. Larvae were dehydrated in an alcohol series and embedded in paraffin and the eyes were sectioned (6 μm thick) and stained with haematoxylin-eosin. The histological sections of the retina were examined under a light microscope (Eclipse 80i; Nikon, Tokyo, Japan) and measurements were made of the distance from the outer edge of the pigment epithelium to the external limiting membrane (v), the width of the pigment epithelium (p) and the length of the cone myoids (m). Then, the retinal indices were computed: p/v for expansion of the pigment epithelium, and m/v for contraction of the cone myoids. Retinomotor responses were quantified as an increase in p/v and a decrease in m/v.
Artemia ingestion experiments
Seabass larvae 10, 15, 20 and 30 days old were tested under light intensities of 0, 0.001, 0.01, 0.1, 1, 10, 100 and 1000 lx (total n = 320 larvae) in the dark-room illumination system described in above. Larvae were starved for 12 h before the experiment, then placed in individual beakers with 300 mL sea water and allowed 90 min to recover from handling stress. For each age group and light intensity, 10 larvae were given 30 Artemia each and allowed to feed for 20 min. Then, MS-222 was added to each beaker, the anaesthetized larvae were removed, and the remaining Artemia in each beaker was counted to determine the number eaten by each larva, later given as Artemia ingestion rates (%). Data were analysed using one-way anova and Tukey's multiple comparison test (SPSS v. 17 Spss Inc, Chicago, IL, USA) .
Larval rearing experiments
Based on the results of the Artemia ingestion experiments, two age groups of Asian seabass larvae were reared in triplicate 5-L aquaria under light intensities of 10, 100 and 1000 lx with illuminated from 0600 to 1800 in a dark room. The first group was 5-day-old larvae stocked at 100 per aquarium, fed rotifers (10 mL−1) at 0800 and 1600, and reared for 10 days (n = 900). The second group was 13-day-old larvae stocked at 50 per aquarium, fed Artemia (1 mL−1) twice a day with some of the Artemia remaining at the next feeding time, and reared for 26 days (n = 450). The seabass larvae evidently fed to satiation. The water in the aquaria was changed every morning by filtered seawater (salinities, 28–30 g L−1). At the end of the rearing experiments, larvae were anaesthetized in 200 mg L−1 MS-222, counted to determine the survival rates, and measured for body weight BW (mg). The BW was measured using an electronic weighing scale after removal of water from body surface by the tissue paper.
Statistical analysis
Statistical analysis was performed using one-way anova. If the one-way anova was significant, the differences between each group were estimated using Tukey's multiple comparison test with the level of statistical significance taken as P < 0.05 (SPSS v. 17).
Results
Eye development
At hatching, seabass larvae had morphologically undifferentiated eyes (Fig. 1a), but the eyes developed soon after hatching (Fig. 1b). The eyes of 2-day-old larvae had well differentiated retinae without rod cells (Fig. 1c).

Retinomotor response under different light intensities
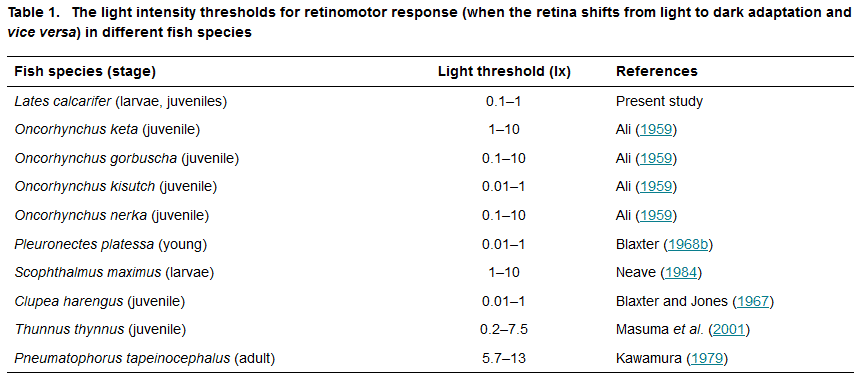
Figure 2 shows retinal sections from seabass larvae in darkness, under low light (0.1 and 1 lx) and under bright light. The cone myoids (m) and the pigment epithelium (p) moved in the absence and presence of different amounts of light. The cone myoids were extended under low light and contracted at high. The pigment layer was relatively thin at low light and markedly thickened at high light.

Figure 3 quantifies the retinomotor responses of three age groups of seabass larvae over eight orders of magnitude of light intensity. The retinae of 10-day-old larvae had almost no retinomotor response, i.e. p/v and m/v hardly changed with light intensity. Retinomotor responses became obvious in 15- and 20-day-old larvae, where p/v markedly increased and m/v markedly decreased at 1 lx (Fig. 3). The light intensity threshold for the retinomotor response was between 0.1 and 1 lx in both 15- and 20-day-old seabass. At 10 lx and brighter, the retinae of 10-, 15- and 20-day-old seabass were almost completely light-adapted and presumably switched to cone vision.
Artemia ingestion rates and feeding thresholds
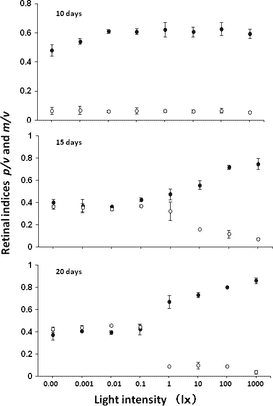
Seabass 10 days old ingested just a few Artemia at 0.1–1 lx and significantly more (92%) at 10 lx and brighter (Fig. 4). Ingestion rates improved with larval age. The ingestion rates of 15-day-old larvae were more than 68% at 1 lx, but only 6–10% at lower intensities. Older seabass 20 and 30 days old ingested Artemia even in the dark (0 lx) and low light (0.001–0.1 lx) with no significant difference (Fig. 4).
Visual thresholds were defined as light intensities required for feeding by vision. The threshold for ingestion of Artemia coincided with the threshold of the retinomotor response (0.1–1 lx) in 20-day-old seabass (Fig. 3). At 10 lx and brighter, the retinae of 10-, 15-, 20- and 30-day-old seabass were almost completely light adapted and presumably detected Artemia by cone vision.
Optimum light intensity for larval rearing
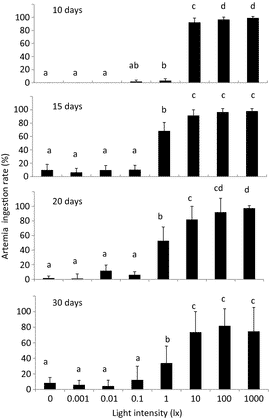
Mean survival rates of seabass larvae reared from 5 to 10 days were 36% at 10 lx, 40% at 100 lx, but only 29% at 1000 lx; however, the differences were not statistically significant (Fig. 5). Seabass reared from 13 to 26 days also showed very similar survival rates (64–73%) under 10–1000 lx, with no statistical difference (Fig. 5). The short rearing periods (5 and 13 days) and the small rearing volume (5 L) may have affected the results, but more likely, the light intensities of 10–1000 lx were within the optimum range for feeding and survival of seabass larvae. The surviving larvae in the younger age group grew best at 100 lx and least at 1000 lx; the older age group grew to very similar body weights under 10–1000 lx (Fig. 6).
Discussion
The retinae of 15- and 20-day-old Asian seabass larvae showed retinomotor responses at a light intensity threshold between 0.1 and 10 lx. The retinomotor responses became more marked with larval growth, but the visual threshold did not change. Rod cells first appear at 10 days in Asian seabass and the retinomotor response first becomes detectable at this time (Yahaya et al. 2011). Asian seabass eggs and early larvae occur in the coastal areas; older larvae move into mangrove swamps during metamorphosis (around 15 days old), and grow there until 20 cm TL (Moore 1982). In general, mangrove estuaries have turbid waters, and a light environment different from coastal waters. Retinomotor responses would help seabass survive in their various habitats.
The thresholds of the retinomotor response vary among fish from 0.01 to 10 lx, with Asian seabass in the middle (Table 1). Seabass 10 days old ingested just a few Artemia at 0.1–1 lx and significantly more at 10 lx. In general, early larvae require at least 10 lx to begin feeding (Tucker 1998). The threshold of Artemia ingestion coincided with the threshold of retinomotor response (0.1–10 lx). Seabass larvae surely detected Artemia by cone vision at more than 10 lx.
Older seabass 15, 20 and 30 days old ingested Artemia even in the dark (0 lx) and low light (0.001–0.1 lx). Under complete dark, seabass probably use sense organs other than the eyes. Seabass larvae have olfactory organs from the yolk sac stage and taste buds in the mouth from 20 days old (Y. Mukai, unpubl. data). They also have free neuromasts arranged on the head surface to receive stimuli from various directions and the number increased with larval growth (Mukai, Chai, Sitti Raehanah & Senoo 2007). These free neuromasts are probably important in the capture of Artemia, which produce vibrations. Free neuromasts play an important role in the detection of zooplankton under dark conditions (Mukai, Yoshikawa & Kobayashi 1994; Mukai & Kobayashi 1995; Mukai 2006).
At light intensities of 10, 100 and 1000 lx, the survival rates of seabass larvae from 5 to 10 days were not significantly different (29–40%); neither were those of larvae from 13 to 26 days (65–73%). Thus, 10 lx is sufficient illumination for larval rearing of both age groups of Asian seabass. In general, fish larvae require at least 10 lx to begin feeding (Tucker 1998). Regarding larval growth, older group fish showed no significant difference, however, younger group showed significantly higher body weight at 100 lx than 10 lx. Fish larvae are reared with phytoplankton or green water (Cobcroft, Pankhurst, Hart & Battaglene 2001). To maintain the phytoplankton, the tanks are usually kept under high light intensities, from several hundred to several thousand lux (Boeuf & Bail 1999), much more than the larvae require for feeding. However, high-density green water actually decreases the light intensity in the rearing water (Cobcroft et al. 2001). Previous study showed that the light intensity in larval rearing tanks 1.8-m deep with Chlorella at 0.2 × 106 cells mL−1 decreases from 1000 lx at the water surface to less than 300 lx at the bottom (Yoseda, Yamamoto, Asami, Chimura, Hashimoto & Kosaka 2008). Therefore, given the physiological sensitivity of the eyes, the feeding efficiency of the larvae and the growth of larvae in the rearing experiments, as conclusion, we recommend that hatchery rearing tanks for Asian seabass be illuminated such that the larvae in the middle layer of fish tank are exposed to approximately100 lx for survival and growth.
May 2014