Identity
Epinephelus coioides Hamilton, 1822 [Serranidae]
FAO Names: En - Orange-spotted grouper, Fr - Mérou taches oranges, Es - Mero de pintas naranjas
Biological Features
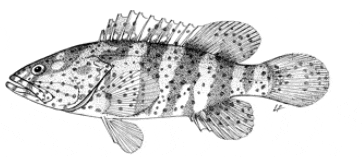
Body elongate, not very compressed; teeth on midside of lower jaw in 2 rows; nostrils subequal; serrae at corner of preopercle moderately enlarged; fourth dorsal spine usually longest; membranes of spinous portion of dorsal fin incised; caudal fin rounded; pelvic fins not reaching anus. Dorsal rays XI, 13-16; anal rays III,8; pectoral rays 18-20; lateral-line scales 58-65; anterior lateral-line scales of adults branched; longitudinal scale series 100-118; scales on body ctenoid except on thorax, abdomen, and a zone above base of anal fin, also narrowing to caudal peduncle; gill rakers 8-10 + 14-17; pyloric caeca about 50.
Light greyish brown dorsally, shading to whitish on side and ventrally, with numerous brownish orange or brownish yellow spots the size of pupil or smaller on head and body; 5 slightly diagonal greyish brown bars on head and body which bifurcate ventrally, the first 4 extending basally into dorsal fin; brownish orange spots on body tend to be arranged in rows parallel to dark bars, this more evident on smaller than larger fish; large dark greyish brown blotches usually present on head, the most prominent behind eye and on opercle; fins whitish to light dusky with brownish orange to brown spots except distally on spinous portion of dorsal fin, caudal fin and pectoral. Unpublished data record maximum weight as 32 kg and length of male at maturity of 120 cm TL.
Images Gallery


Profile
Historical Background
Groupers are generally cultured in floating net cages or earthen ponds, but cage culture is more common in Southeast Asia. In 1979, the Penghu Station of the Taiwan Fisheries Research Institute (TFRI) started artificial propagation by using hormone-inducing technique. E. coioides is one of the two major cultured groupers in Taiwan P C. The National Mariculture Center, Bahrain has conducted mass fry production trials of this species since 1992. Grouper pond production is becoming an attractive alternative to intensive shrimp culture in countries where management problems have forced growers to abandon shrimp farming. This species has been tested in several countries as a potential species for mariculture.
Main Producer Countries
The map shown below is constructed from FAO reported statistics for this species. Farming activities also occur in other countries including China, Thailand, Taiwan P.C., Indonesia, and are reported to FAO in the generic category "Grouper unidentified".
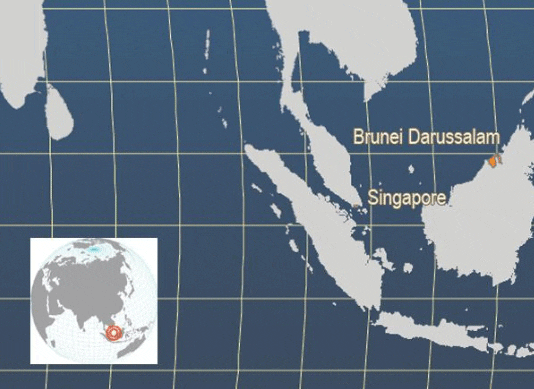
Main producer countries of Epinephelus coioides (FAO Fishery Statistics, 2006)
Habitat and Biology
Epinephelus coioides occurs in the western Indian Ocean from the southern Red Sea to Natal and east to the western Pacific where it is distributed from Ryukyu Islands to New South Wales. It ranges east into Oceania only to Palau in the Northern Hemisphere and Fiji in the Southern. Orange-spotted groupers inhabit turbid coastal reefs and are often found in brackishwater over mud and rubble. Juveniles are common in the shallow waters of estuaries over sand, mud and gravel and among mangroves, feeding on small fish, shrimp, and crabs. E. coioides are eurythermal and euryhaline.
The major spawning period is March to June. Females mature at 250-300 mm TL at an age of 2-3 years; sexual transition occurs at a total length of 550-750 mm. Fecundity estimates varied from 850 186 ova in a 350 mm TL fish to 2 904 912 ova for one of 620 mm TL. Eggs are pelagic; best survival of larvae is attained at 30 °C and 30 ‰. Successful artificial propagation of E. coioides has been reported in Malaysia. They probably spawn during restricted periods and form aggregations when doing so and the eggs and early larvae are probably pelagic.
Production
Production Cycle
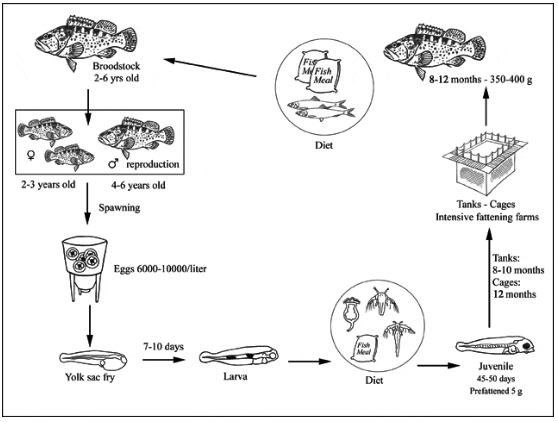
Production cycle of Epinephelus coioides
Production Systems
Seed Supply and Hatchery Techniques
Brooders of E. coioides are stocked in separate 50 m3 tanks. Most of the brooders are collected from the wild and reared for 1 to 7 years using seawater at a constant temperature of 27-28 °C and 45 ‰, following standard culture methods. The fish are fed with frozen sardines, mackerels, cuttlefish, squids and top-shell clams because these foods have high contents of cholesterol, phospholipids and polyunsaturated fatty acids. Artificial diets containing hormones used for maturation or sex-reversal are also used.
Natural Spawning
At the National Mariculture Center in Bahrain orange spotted grouper eggs are collect following standard methods. Under a constant temperature regime of 27-28 °C, natural spawning was maintained during a 33 month period between October 1992 and July 1995 and the total number of eggs collected during that period was 279 million. Daily floating egg rates varied from 5.6 to 69.6 per cent (average 36.8 percent). Spawning relates to lunar rhythm: the quantity of spawned eggs is high about one week before and after the new moon, and is low or none near the full moon. Under ambient temperatures, spawning occurs in April and May.
Induced Spawning
Whenever brooders do not spawn naturally, mature females and males are selected from broodstock tanks and injected with Human Chorionic Gonadotropin (HCG) at 700 and 500 IU/kg BW respectively. After the injection, the fish are stocked at 28.0-28.5 °C in a 50 m3 tank. Brooders usually start spawning between 36 hrs and 48 hours after injection. There were over ten grouper breeder farms raising >10 000 spawners in Southern Taiwan P.C. in 1998; these were producing 40 billion eggs/yr.
Management of eggs and newly hatched larvae
Eggs are collected from the egg collection net within 3 hours after spawning. Floating eggs are stocked at 600 000 per 1 m3 tank. The eggs are incubated in the same tank with moderate aeration and running water until they hatch. Newly hatched larvae are collected from the water surface by glass beakers within 3 hours after hatching and their number estimated by collection in a 20 L bucket and sub-sampling. Larvae are then transferred to larval rearing tanks.
Larval Rearing
Larval rearing is conducted using 20 m3 concrete tanks (4 x 2.5 x 2.5 m (HxWxD). Standard methods for larval rearing are applied. Artificial lighting is provided as follows [hatching day = day 0]: at day 3 when larvae start feeding, from 07:00 to 24:00; at days 4 and 5, continuous; at day 6, from 07:00 to 22:00; at day 7, 07:00 to 20:00. Natural lighting is provided from Day 8 onwards only.
Larvae start feeding immediately after the mouth opens. Newly open-mouth larvae are fed once a day with enriched Brachionus plicatilis, S-rotifer of 160-180 µm size at a density of 5-6 rotifers/ml. At 3-20 mm TL, larvae are fed twice daily with S-rotifer of 180-200 µm at 5-10 rotifers/ml. Larvae of 5 to 8 mm, 8 to 20 mm and larger than 20 mm TL sizes are fed almost continuously with microfeeds of 140-410 µm, 315-580 µm and 479-800 µm, respectively. At TL 6-25 mm, the larvae are also fed enriched Artemia nauplii to satiation for 1 to 3 hrs once in the afternoon.
Nursery
There are two production systems used for the nursery stage - indoor and outdoor. The indoor system uses tank sizes of 30-50 m3 while the outdoor system uses ponds of >200 m3.
Outdoor System
Hatchery reared or wild-caught fry are nursed in tanks or hapa nets until they reach 6 cm. Hapa nets [1 x 2 x 1.5 m; -2 mm mesh] are set in tanks or ponds or inside floating net cages and stocked
Indoor System
Nursery tanks vary from 30 to 50 m3 in either semi-intensive or intensive flow-through systems. Such tanks are stocked with 3. Higher densities of over 1 000/m3 are sometimes used in flow-through or recirculation water systems. The fish are graded every 5-7 days until they reach >6 cm after 45-60 days. At this stage, fish are transferred to grow-out ponds or floating cages.
Ongrowing Techniques
Earthen Pond systems
Ponds are prepared and fertilized. Once natural food is abundant, adult tilapia are added at a stocking density of 5 000-10 000/ha to produce fingerlings to serve as live prey for the groupers. Grouper fingerlings (~6 cm TL) are added at 5 000-10 000/ha at least a month after the release of adult tilapia. Sorting and grading of the fingerlings is carried out weekly to prevent cannibalism and to minimize competition for space and food. If tilapia fingerlings are not abundant, supplementary feeding is carried out using chopped fish at 5 per cent BW/day, half early in the morning and the rest late in the afternoon. When the fish weigh about 200 g, feeding is reduced to once daily with fresh or frozen chopped fish at 5 per cent BW or with pellets at 2 per cent BW. 20-50 per cent water exchange takes advantage of spring tides or pumped water from reservoirs and is carried out at least twice/week. Paddlewheel aerators are used when DO2 falls below 4 ppm. Water quality is maintained at pH 7.5-8.3, 25-32 °C, 20-35 ‰, 4-8 ppm DO2; 2-N) and 3-N.
Floating Net Cage Systems
Net cages (8 mm mesh) are used for 2-10 cm fingerlings; 25 mm mesh is used for larger fish. Grouper fingerlings are stocked at 15-20/m3. Grading is carried out at least once a month. Fish are fed with appropriate fresh or frozen chopped fish daily at 10 per cent BW or with pelleted feeds 3 per cent BW, half early in the morning and half late in the afternoon. 0.5 per cent vitamin and mineral premix is added to the properly thawed trash fish before feeding. Floating net cages should be moved to a new site every 2-3 years of culture to allow deteriorating bottom conditions to recover. The duration of culture in the grow-out phase is 4-7 months, depending on the preferred size at harvest.
Feed Supply
Generally, farmers depend on commercial suppliers for grow-out feeds. Costs for groupers vary from USD 1 100 to 1 200/tonne (2010).
Harvesting Techniques
Groupers are harvested at 400 g or above, depending on specific market demand. Fish are marketed either live for the restaurant business and international markets or freshly caught for local markets.
Harvesting in Earthen Ponds
Fish are harvested by seine in the early morning or late afternoon. The water is disturbed (agitated) two hours before harvest to prevent occurrence of rigid muscles in the fish. Alternatively, for partial harvesting, modified lift nets may be installed in feeding areas; these have to be lifted slowly to avoid fish escape. It is advisable to install 8x2x1.5 or 8x4x1.5 m net cages (25 mm mesh) in the ponds to hold fish temporarily. The stocking density in these holding cages should not exceed 20/m3.
Harvesting in Net Cages
Feeding is stopped 1-2 days before marketing. The net cage should be inspected for any damage and then lifted slowly from one side to concentrate the fish in one corner. A fine, soft knotless scoop net or soft plastic screen is used to catch the fish. Care must be taken to avoid loss of scales or causing lesions on fish during harvesting.
Handling and Processing
Harvested fish are stocked in an aerated conditioning tank. To slow metabolic activity, the water temperature is slowly lowered to 20 °C (2-3 °C/hr) by adding crushed ice in plastic bags or by using cooling pumps. 3-5 fish are stocked into each 20x20x30 cm double-lined plastic bag. Biomass is limited to 2-3 kg/bag. Oxygen is added at a 1:3 water:oxygen ratio before sealing with a rubber band. The bags are packed in styrofoam boxes to which an adequate amount of frozen gel packs, ice bags or frozen water in sealed plastic bottles wrapped in old newspapers is added to keep the temperature low during transportation. This method is suitable for air transport for a period not exceeding 8 hours after packing.
Production Costs
Fry usually comprise between 15-20 per cent of ongrowing costs. In hatcheries, labour costs are 20 to 25 per cent of the total. Other large factors are feed (30 percent), followed by administrative expenses and fuel and power requirements for heater/chillers and automatic feeders. Generally, ongrowing costs are lower in larger-scale farms.
Diseases and Control Measures
| In some cases antibiotics and other pharmaceuticals have been used in treatment but their inclusion in this table does not imply an FAO recommendation. | ||||
|---|---|---|---|---|
| Disease | Agent | Type | Syndrome | Measures |
| Blister disease | Iridovirus GIV-2 | Virus | Highly localised severe inflammation of epidermal and dermal layer; dermis necrotized, containing exudation and haemorrhagic infiltration at the area of intact layer; presence of icosahedral to round-shaped virions with a diameter of 180-200 nm in infected liver, spleen, kidney and lesions | Good prophylaxis & good husbandry conditions |
| Vibriosis | Vibrio spp. | Bacteria | Significant numbers of monogenean parasites causing gill lesions observed | Vaccination, good prophylaxis & good husbandry conditions |
| Parasitic Infestations | Cryptobia sp, Scyphidia sp. Vorticella sp. Dactylogyrus sp. Neobenedenia girellae Gnathia sp. | Protozoans Monogeneans Isopod | Skin and gill lesions; depigmentation; ulceration; skin area haemorrhages | Good prophylaxis & good husbandry conditions |
| Swim-Bladder Syndrome | Undiagnosed or unknown | Over-inflation of the swim bladder; loss of buoyancy control | Good prophylaxis & good husbandry conditions | |
| Popeye (Exopthalmosis) | Undiagnosed or unknown | Extremely bulging eyes | No known successful treatment | |
Suppliers of Pathology Expertise
Assistence can be obtained from the following sources:
- Penghu Station, Taiwan Fisheries Research Institute (TFRI), Taiwan P.C.
- Japan International Cooperation Agency, Japan.
- Aquatic Animal Health Research Institute, Department of Fisheries, Kasetsart University Campus, Jatujak, Bangkok, Thailand.
- Gondol Research Institute for Fisheries of Indonesia, Bali, Indonesia.
- National Mariculture Center, Directorate of Marine Resources, Bahrain.
Statistics
Production Statistics
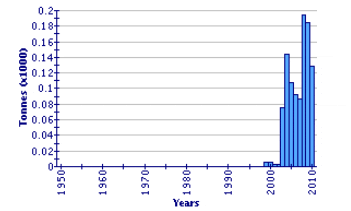
Global Aquaculture Production of Epinephelus Coioides
(FAO Fishery Statistic)
Market and Trade
The wholesale price of live market-size groupers was about USD 12.99/kg in May 2010 in Hong Kong and USD 15.96/kg in February 2010 in Bahrain. In the year 2000, market demand for cultured groupers was about 20 000-25 000 tonnes. Since then production has been steadily rising with increasing global demand. Generally, groupers are a popular food fish and it is estimated that the market demand may reach 100 000 tonnes per year in 2020. Therefore, sustainable aquaculture of groupers and their related species deserves further development. Restaurants display live groupers in aquaria fitted with water recirculation systems.
As production techniques have improved and off-flavours have been controlled in Taiwan P.C., Singapore and Japan by keeping the market-sized fish in tanks with running water for two days without feeding, orange-spotted groupers have moved into the mainstream seafood markets of developed countries. In highly industrialized countries, small markets for live groupers or frozen imports developed among immigrant communities. With the appearance of fresh grouper fillets from tropical countries, new markets opened in upper echelon restaurants, casual dining restaurant chains, hypermarkets and discount stores. Virtually all casual dining restaurant chains in the Orient feature groupers, which are an ideal addition to the menu due to their reasonable price, year-round supply, mild, delicious flavour and flexibility in preparation. China, a major exporter of groupers, has great potential for market development to supply a rapidly growing middle class.
Status and Trends
Groupers have become one of the most important aquaculture and traded commodities in the Asia-Pacific region. It is also an important fish in the livelihoods of small and large-scale coastal fish farmers. Groupers are considered as a high-value species with a high potential for contributing to the economic development of these countries. The expanding trade in live groupers of various ages and stages, whether for aquaculture or for seafood restaurants, has increased demand since 2006.
Orange-spotted grouper, E. coioides is one of the few species among farmed grouper species. The development of new faster-growing strains through selective breeding techniques and use of Intensive cost-effective recirculation systems are imperative to increase the production.
Main Issues
The main issues constraining the further development of orange spotted grouper farming include:
- This species grow more slowly on formulated feeds with high protein levels than many carnivorous farmed species.
- Although grouper culture is widespread in Asia and the Pacific, its continued development is constrained by the limited availability of fingerlings. Most economies, with the exception of China Taiwan P.C., rely almost totally on wild-caught fry and fingerlings for stocking.
- The inadequate supply of seed is further aggravated by lack of appropriate handling techniques during collection, transport and storage of collected fish, and sometimes by an unregulated management of the wild stocks.
- There is a lack of appropriate techniques for efficient grouper culture to marketable sizes. A major production constraint is heavy mortality of groupers during the collection and culture phases due to handling stress and diseases. High mortality rates occur in the early larval stages and larval cannibalism is rampant. Farmed fish are also often prone to diseases (especially viral diseases and parasitic infestations). Research to solve these problems is under way in Japan, Taiwan P.C., Thailand and Bahrain.
- Used during production mainly to prevent and treat bacterial disease, antibiotics are leading to the development of antibiotic-resistant bacteria that are pathogenic to humans. The development of antibiotic resistance by pathogenic bacteria is considered to be one of the most serious risks to human health at the global level. Many countries around the world have introduced, changed or tightened national regulations on the use of antibiotics, in general and within the aquaculture sector.
Responsible Aquaculture Practices
Responsible aquaculture at the production level should be practised in accordance with the main principles of environmental and ecological protection – see Article 9 of the FAO Code of Conduct for Responsible Fisheries.
Integrated efforts could help to explain the development of intensive productions systems and answer questions raised by the public. Veterinarians have the responsibility for the health assessment of reared animals; they control and measure the state of health with the aim of preventing disease. Thus they are the most suitable people to interact with farm operatives and government inspectors in examining the animals and deciding whether or not they are showing signs of good health and well-being. Animal health in this sense would aim to create equilibrium between the animal and its new environment in intensive conditions.
The utilization of non-destructive devices for grouper collection, proper fish handling and increased efficiency in culture management could create socio-economic and environmental benefits. A well-developed grouper culture operation, complemented by appropriate wild grouper fishery management could provide sustainable employment for many people – from marginal fishers to farmers to traders.
The demand for wild seed has led to unsustainable and illegal practices such as the use of cyanide to capture large numbers of seed with relatively low investment in time and effort. Grouper fisheries that are based on illegal or destructive fishing practices underline the urgent need for habitat protection and sustainable utilization of natural resources.
References
Bibliography
Al-Baharna, W.S. 1986. Fishes of Bahrain. Directorate of Fisheries, Bahrain. 294 pp.
Boulenger, G.A. 1895. Catalogue of the Fishes in the British Museum. 2 ed. Vol. 1. British Museum (Natural History), London. xix + 391 pp.
Chong, Y.G. & Chao, T.M. 1986. Common diseases in marine finfish. Primary Production Department, Republic of Singapore. 34 pp.
Doi, M., Munir, M.N., Razali, N.L. & Zulkifli, T. 1991. Artificial propagation of the grouper, Epinephelus suillus at the marine finfish hatchery in Tanjong Domeng, Terengganu, Malaysia. Kertas Pengembangan Bil. 167, Department of Fisheries, Malaysia, 41 pp.
Gunther, A. 1859. Catalogue of the acanthopterygian fishes in the collection of British Museum. Vol.1, British Museum, London. xxxi+524 pp.
Hamilton, F. 1822. An account of the fishes found in the river Ganges and its branches. Archibald Constable and Company, Edinburgh. 405 pp.
Heemstra, P.C. & Randall, J.E. 1986. Serranidae (Subfamilies Epinephelinae and Serranidae). In: W. Fischer & G. Bianchi (eds), FAO Species Identification Sheets, Western Indian Ocean. Vol. 4, 241 pp. FAO, Rome.
Hua, D.K., Cai, M.L. & Zhang, Z.Y. 1994. Studies on the grouper’s syndrome: I. Etiology and histopathology. In: L.M. Chou, A.D. Munro, T.J. Lam, T.W. Chen, L.K.K. Cheong, J.K. Ding, K.K. Hooi, H.W. Khoo, V.P.E. Phang, K.F. Shim & C.H. Tan (eds), Proceedings of the Third Asian Fisheries Forum, 26-30 October 1992, Singapore, pp. 353-356. Asian Fisheries Society, Manila, Philippines.
Kawahara, S., Al-Bosta, A.A. & Shams, A.J. 1996. Hatchery production of grouper, Epinephelus coioides, and rabbitfish, Siganus canaliculatus, at the National Mariculture Center, Bahrain: 1995. National Mariculture Center, Directorate of Fisheries, Ministry of Works & Agriculture, State of Bahrain. 19 pp.
Kawahara, S., Al-Bosta, A.A. Al-Murbaty, F.A. & Shams, A.J. 1995. Hatchery production of the grouper, Epinephelus coioides, at the National Mariculture Center, Bahrain: 1993-1994. National Mariculture Center, Directorate of Fisheries, Ministry of Works & Agriculture, State of Bahrain. 22 pp.
Lavilla-Pitogo, C.R., Castillo, A.R. & de la Cruz, M.C. 1992. Occurrence of Vibrio sp. infection in grouper (Epinephelus suillus). Journal of Applied Ichthyology, 8:175-179.
Mathews, C.P., Samuel, M. & Baddar, M.K. 1986. Sexual maturation, length and age in some species of Kuwait fish related to their suitability for aquaculture. Kuwait Bulletin of Marine Science, 8:243-256.
Playfair, R.L. & Gunther, A.C.L. 1867. The fishes of Zanzibar. John van Voorst, London. (Reprint edition 1971, Newton K. Gregg, Kentfield, California). xiv + 153 pp.
Randall, J.E. & Heemstra, P.C. 1991. Indo-Pacific fishes. Revision of Indo-Pacific groupers (Perciformes: Serranidae: Epinephelinae), with descriptions of five new species. Indo-Pacific Fishes, No 20. 332 pp. B.P. Bishop Museum, Honolulu, Hawai’i.
SEAFDEC. 2001. Husbandry and health management of grouper. Asia-Pacific Economic Cooperation Fisheries Working Group 01/2000. Aquaculture Department, Southeast Asian Fisheries Development Center, Manila, Philippines. 94 pp.
Weber, M. & de Beaufort, L.F. 1931. The fishes of the Indo-Australian Archipelago. Vol. 6. E.J. Brill, Leiden. xii.+448 pp.
October 2012




