 Figure 1: Optimal injection point. The fish shall be injected in the abdominal midline, slightly in front of the pelvic bone (that lies beneath the flesh) and one fin length in front of the base of the pelvic fins (marked with an X). The green area depicts the ideal area for injection and the yellow area depicts the outer limits for acceptable deviations to this. |
Recent experience in the field has shown that the quality of the vaccination procedure per se may have a big influence on how the fish progresses through the whole production cycle, in terms of both the efficacy of the vaccine to protect against the targeted diseases and any side effects that may arise from the vaccination.
The most serious problems often arise when the S0-smolts are being vaccinated and when a large number of fish are being vaccinated during a short period. Under these circumstances, the quality of the vaccination procedure per se may be reduced and a higher proportion of the fish are of a small size, which in turn increases the risk of incorrect vaccination. Indeed, even apparently insignificant deviations from the recommended injection point can reduce the vaccine efficacy and also result in a higher incidence of side effects (i.e., local reactions in the internal viscera).
Over the years, Intervet Norbio AS has performed several vaccination checks in Norway. There has certainly been an improvement in the vaccination technique but there is still room for additional improvements. The greatest challenge is to perform the vaccination correctly on every fish on every day of the vaccination period.
It is also worth mentioning that new legislation entered into force in Norway in December 2004 (Norwegian Regulation of Aquaculture Management, § 55). The new legislation requires closer inspection of the vaccination process when automatic vaccination equipment is used. [It is also possible that similar legislation will be enacted in other fish vaccination countries in the future.]
Recommended placement of the injection point
 Figure 2: Adjustment of the length of the needle. The needle should penetrate 1 to 2 mm into the abdominal cavity. |
The recommended position of the injection point is in the midline of the abdomen, one pelvic fin length in front of the base of the pelvic fins.
Intervet Norbio now has extensive research data showing that water temperature and fish size are key factors that may influence the development of local reactions in vaccinated fish. Optimally, the temperature should be below 15°C from the time of vaccination until several weeks after sea transfer, and the individual fish size at vaccination should be ≥35 grams. High water temperature and/or a sudden increase in water temperature during this window will possibly contribute to a greater incidence and severity of local reactions in the fish. Although it is likely that some of the link between (small) fish size and greater side effects is due to fish size per se, undoubtedly some is also due to operators injecting the vaccine in the wrong position in fish that are below the recommended size.
Deviation in the point of injection
Section 55 of the Norwegian Regulation of Aquaculture Management states that the percentage of deviation from the correct point of injection shall not exceed 0.1 %. However, in practice, it will be hard to attain such a low percentage deviation but the rule of thumb should be to keep the deviation as close as possible to 0.1%.
What do we mean by deviation in injection point?
 Figure 3: The injection point is placed too far forward, resulting in injecting inside the pyloric caecae (may also occur if a vaccinator forces the needle). |
A deviation in the injection point occurs when the vaccine is deposited in a way where it does not float freely in the abdominal cavity. When this occurs, it usually means that the injection point was outside the recommended injection area. Other reasons could be that the depth of the needle was not correct (Figure 2, adjustment of the length of the needle) or that the angle of the needle was not optimal. These types of injections can lead to improper vaccination and/or unwanted damage.
Examples of deviations in the injection point are:
Incorrect position of the injection point (Figure 3), i.e., the injection point is placed outside the yellow area depicted in Figure 1.
Too shallow an injection (Figure 4). The vaccine is injected directly into the muscle or between the muscle (abdominal wall) and the peritoneum.
Too deep an injection (Figure 5). The vaccine is injected into the internal organs; for instance, into the appendix (pyloric caecae), intestines or kidney.
Tears and bleeding at the injection point (Figure 6). The needle causes wounds in the skin/muscle on the outer surface or on the inside of the abdominal wall.
Wrong timing of injection. The vaccine is injected when the needle is on its way into or out of the abdominal cavity. Usual causes of deviation of the injection point
Uneven size of fish. If the difference between the small and big fish in the group to be vaccinated is too great, this could lead to either shallow or deep injections.
Lack of validation of the vaccination technique. The deviation in the injection point during the vaccination period will not be recognised and corrected.
The speed of vaccination is too high. This could lead to reduced precision of vaccination.
Lack of routine adjustment of the equipment for each individual vaccinator, together with long working days. This creates a higher risk for human error with vaccination.
Not with-holding feed for a period before vaccination increases the risk of injecting the vaccine into the internal organs.
Vaccination with improperly honed needles or needles that are blunt. This leads to tears and bleeding at the injection point.
 Figure 4: Vaccination with a needle that is too short or placement of the injection point too far back. The vaccine is deposited between the peritoneum and the abdominal wall. |
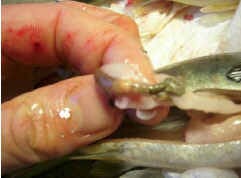 Figure 5: Depositing the vaccine into the intestine raises the danger of a reduced vaccine efficacy. |
Consequences of a deviation from the recommended injection point:
Mortality after vaccination. Injection of the vaccine into internal organs may lead to acute mortality.
Reduced efficacy. Injection of the vaccine into the muscle or internal organs, or incomplete injection of the correct vaccine volume, can result in insufficient protection against diseases after vaccination.
Side effects (local reactions). Injecting the vaccine into the pyloric caecael region or into the stomach/intestines can lead to a traumatic local inflammatory reaction as a result of leakage of contents from the intestines into the abdominal cavity.
Downgrading of fish quality at harvest as a result of pigmentation/ melaninisation in the muscle. Injecting vaccine directly into the external muscle or between the membrane (peritoneum) covering the inner side of the abdominal cavity and the abdominal wall (Figure 3).
"Unvaccinated" fish. Where the vaccine has been deposited inside the front or hind intestine, or inside the stomach. The vaccine would then be flushed out with the faeces a short time after vaccination. When a high percentage of fish are vaccinated in this way, there is the additional risk that the group protection (or "herd immunity") will not take place.
Summary
 Figure 6: Tears and bleeding at the injection point due to using damaged or blunt needles, or by too rough handling. It raises the danger of vaccine leakage or wounds that can lead to infection. |
Correct placement of the injection point is best attained when the fish to be vaccinated are equal in size and when the vaccination is performed by trained personnel using good work practices. The vaccination should be performed in co-operation with the fish health service and the vaccine manufacturer. Routine checking of the vaccine procedure is crucial for attaining a consistently good quality vaccination process. Correct vaccination of fish ≥35 grams in size and at water temperatures below 15°C with a quality vaccine will normally result in fish with both good protection against disease and a low incidence and severity of side effects. This will ensure the farmer gets good production performance and correspondingly good margins. Importantly, it also means that the farmer can ensure good animal welfare standards according to the Norwegian Regulation of Aquaculture Management which is founded on the Norwegian Law of Animal welfare.
Source: Intervet International B.V - December 2005



