Hence, selection criteria of probiotics for aquaculture should be based on their antagonism towards pathogens (through competitive exclusion), their growth, attachment to intestinal mucus and production of beneficial compounds (Vine et al., 2004). Since fish and shrimp are cultured under different conditions the optimum temperature range should also be considered for selection of the right probiotic strain. Another important factor is that probiotics always lack pathogenicity. After all, probiotics should contribute to efficient production in a sustainable way, promoting healthy and robust animals (Brittain et al., 2002).
Lactic acid bacteria (LAB) are potential probiotic candidates in aquaculture and are also known to be present in the intestine of healthy fish. Enterococcus faecium is one of the most commonly used lactic acid producing bacteria in animal nutrition and has become a focus of attention for use in commercially farmed aquatic species. Enterococci are Gram-positive, facultative anaerobic bacteria which are widely distributed in nature and considered as bacteria of low pathogenicity (Klare et al., 2003). Probiotic enterococci infections haven´t been reported in the veterinary medicine, so the risk appears to be limited (Rinkinen et al., 2003). Eaton and Gasson (2001) found that E. faecium strains were also generally free of virulence determinants. Concerning possible acquired antibiotic resistances, testing the antibiotic profile of enterococci used as probiotics is of relevance. However, these antibiotic resistances are species- or genus-specific properties (Klare et al., 2003). Once the probiotic enterococci are accurately identified, they can be tested for specific resistances.
Several important ways in which probiotics producing lactic acid (such as E. faecium) can provide a performance benefit are improving intestinal microbial balance (Fuller, 1989), stimulating the immune system and decreasing pH as well as the release of bacteriocins (Rolfe, 2000). Bacteriocins are small peptides which are characterized by their ability to inhibit pathogenic bacteria; whereas, some have a narrow spectrum of activity while others inhibit a wide variety of bacteria.
The use of the bacteriocin-producing E. faecium with probiotic properties alone or in combination with other beneficial intestinal bacteria was investigated in different studies.
In vitro studies using the agar spot method (Rosskopf, 2010) have shown that Enterococcus faecium (strain IMB 52) has inhibition properties against a wide spectrum of aquatic pathogens including Yersinia ruckeri, Vibrio harveyi, Streptococcus agalactiae and Aeromonas veronii. Similar observation was made by Swain et al. (2009) who proved the inhibitory activity of E. faecium isolated from brackishwater fish against V. harveyi and V. parahaemolyticus. This demonstrates the potential applications of E. faecium from fish intestine for controlling pathogenic vibriosis in shrimp culture. It has also been reported that survival rates of European eels (Anguilla Anguilla L.) fed with E. faecium were significantly higher than in the control groups after challenged with Edwardsiella tarda (Chang and Liu, 2002).
Wang et al. (2008) demonstrated that the addition of E. faecium (1 x 107 CFU/mL) in aquaria water could significantly increase final weight and daily weight gain (DWG) of tilapia. Since certain immunological parameters (myeloperoxidase and respiratory burst activity) of tilapia were improved as well, the increased growth performance might be attributed to less bacterial challenge, confirming the benefit for the non-specific innate immunity of this kind of fish.
Panigrahi et al. (2007) examined immune modulation including cytokine gene expressions of rainbow trout (Oncorhynchus mykiss) and demonstrated that these parameters were improved by probiotic feeding of freeze-dried Lactobacillus rhamnosus, Enterococcus faecium or Bacillus subtilis (109 CFU/g) after 45 days. Particularly the fish fed the E. faecium strain showed better performance which could possibly be linked to the suitable ambient temperature conditions of this strain. Temperature is a major environmental factor controlling microbial growth and the ideal conditions differ among microorganisms. E. faecium was found to be more psychrotolerant than the other two bacteria, growing well at temperatures ranging from 12 to 30 °C. Rosskopf (2010) showed that the optimum temperature range for E. faecium (strain IMB 52) reaches as far as to 37 °C.
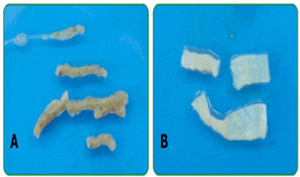
B) Intestinal epithelium without faecal material
Using the fluorescence in situ hybridization (FISH) technique, Supamattaya et al. (2005, 2006) have demonstrated in a series of in vivo studies that E. faecium (strain IMB 52) is also able to populate the intestine of white shrimp (Litopenaeus vannamei) and Nile tilapia (Oreochromis niloticus) (Picture 3 and Table 1) and induce a positive impact on bacterial ecology of the gut by inhibiting Vibrio spp. through competitive exclusion. E. faecium was even detected in the fish gut and faeces 10 days after product administration. In addition, it was observed that dietary application of E. faecium (strain IMB 52) alone or in combination can improve growth performance, enhance the immune response (by increasing granular hemocytes level) (Supamattaya et al., 2005) and increase survival rate in shrimp contaminated with Vibrio parahaemolyticus (Krummenauer et al., 2009).
E. Faecium in Tilapia's Intestine and Faeces at Day 1 and Day 10 After Stopping to Feed Probiotics Including this Probiotic Strain
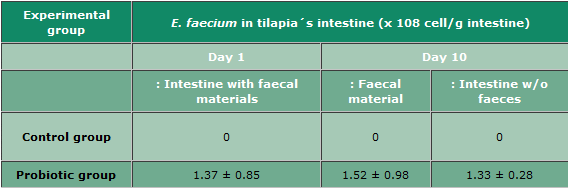
Means ± SD. Using Fluorescent in situ hybridization (FISH), 5 - 15 microscopic fields were counted.
From these studies it was concluded that Enterococcus faecium (strain IMB 52) is a safe and promising probiotic candidate for aquatic species which can be applied in different combinations.
Conclusion
Maintaining the balance of critical parameters and effective disease control remain fundamental requirements for successful aquaculture. In order to withstand the high stocking densities in shrimp and fish production probiotics are a promising feed additive to stimulate animal growth and advance disease resistance. Enterococcus faecium as probiotic strain in aquatic species is increasingly recognized as safe and can be applied in different combinations. E. faecium can grow at a wide range of temperatures and thus has an advantage over other bacteria considering that fish and shrimp are reared at different temperatures and conditions.
The current paper illustrated with in vitro studies and performance trials the potential benefits of E. faecium as probiotic feed additive for fish and shrimp, either by stimulating the development of a healthy gut microflora or by inhibiting pathogenic bacteria like Vibrio spp., Yersinia spp. and Aeromonas spp.
References
Brittain A, Lee CS, O´Bryen PJ (2002) Workshop discussion and recommendations on “Microbial contributions to aquatic nutrition”. In: Microbial approaches to aquatic nutrition within environmentally sound aquaculture production systems. The World Aquaculture Society, United States.
Chang CI, Liu WY (2002) An evaluation of two probiotic bacterial strains, Enterococcus faecium SF68 and Bacillus toyoi, for reducing edwardsiellosis in cultured European eel, Anguilla Anguilla L. Journal of Fish Diseases 25 (5): 311 – 315.
Eaton TJ, Gasson MJ (2001) Molecular screening of Enterococcus virulence determinants and potential for genetic exchange between food and medical isolates. Appl. Environ. Microbiol. 67: 1628 – 1635.
Fuller R (1989) Probiotics in man and animal. Journal of Applied Bacteriology 66: 365 – 375.
Klare I, Konstabel C, Badstübner D, Werner G, Witte W (2003) Occurrence and spread of antibiotic resistances in Enterococcus faecium. International Journal of Food Microbiology 88: 269 – 290.
Krummenauer D, Abreu PC, Lara G, Poersch L, Encarnacao P, Wasielesky Jr W (2009) The Effect of Probiotic in Litopenaeus vannamei Biofloc Technology Culture System contaminated with Vibrio parahaemolyticus. Abstract World Aquaculture Conference, Mexico.
Panigrahi A, Kiron V, Satoh S, Hirono I, Kobayashi T, Sugita H, Puangkaew J, Aoki T (2007) Immune modulation and expression of cytokine genes in rainbow trout Oncorhynchus mykiss upon probiotic feeding. Developmental and Comparative Immunology 31: 372 – 382.
Rinkinen M, Jalave K, Westermarck E, Salminen S, Ouwehand AC (2003) Interaction between probiotic lactic acid bacteria and canine enteric pathogens: a risk factor for intestinal Enterococcus faecium colonization? Veterinary Microbiology 92: 111 – 119.
Rolfe RD (2000) The Role of Probiotic Cultures in the control of Gastrointestinal Health. Symposium: Probiotic Bacteria: Implications for Human Health. The Journal of Nutrition: 396S - 402S.
Rosskopf S (2010) In vitro Evaluation of Inhibitory Activities of Probiotics against Aqua-pathogens for their possible Use in Aquacultures. Bachelorthesis, Biomin Holding GmbH, Tulln.
Supamattaya K , Viriyapongsutee B, Ruangsri J, Encarnacao P, Schatzmayr G (2005) Effect of probitoic Enterococcus faecium and Phycophytic Substances on Growth Performance and Health Condition of White Shrimp (Penaeus vannamei).
Swain SM, Singh C, Arul V (2009) Inhibitory activity of probiotics Streptococcus phocae PI80 and Enterococcus faecium MC13 against Vibriosis in shrimp Penaeus monodon. World J Microbiol Biotechnol 25: 697 – 703.
Vine NG, Leukes WD, Kaiser H (2004) In vitro growth characteristics of five candidate aquaculture probiotics and two fish pathogens grown in fish intestinal mucus. FEMS Microbiology Letters 231: 145 - 152.
Wang YB, Tian ZQ, Yao JT, Li WF (2008) Effect of probiotics, Enterococcus faecium, on tilapia (Oreochromis niloticus) growth performance and immune response. Aquaculture 277: 203 – 207.
April 2012

