Introduction
When feed is eaten by fish it is metabolized into the energy, nutrients, and proteins used for survival and growth. As with all animals, there is waste produced by these normal metabolic processes. Ammonia is the principal waste product excreted by fish. In trace amounts, ammonia is odorless and colorless, so the only way for an aquaculturist to know if it is there is to test the water.
Of all the water quality parameters which affect fish, ammonia is the most important after oxygen, especially in intensive systems. In small amounts, ammonia causes stress and gill damage. Fish exposed to low levels of ammonia over time are more susceptible to bacterial infections, have poor growth and will not tolerate routine handling as well as they should. Ammonia is a killer when present in higher concentrations, and many unexplained production losses have been caused by ammonia.
In water, ammonia occurs in two forms, which together are called the Total Ammonia Nitrogen, or TAN. Chemically, these two forms are represented as NH4+ and NH3. NH4+ is called Ionized Ammonia because it has a positive electrical charge, and NH3 is called Unionized Ammonia since it has no charge. This is important to know, since NH3, unionized ammonia (abbreviated as UIA), is the form which is toxic to fish. Water temperature and pH will effect which form of ammonia is predominant at any given time in an aquatic system.
The Nitrogen Cycle
In aquaculture, a mechanism called The Nitrogen Cycle eliminates ammonia from the water. As ammonia is excreted, it is converted to another compound called Nitrite (NO2). Nitrite is also toxic to fish and is discussed in a separate publication. This change from ammonia to nitrite is accomplished by bacteria called Nitrosomonas. Another group of bacteria, called Nitrobacter, convert nitrite to Nitrate (NH3). Nitrate is not toxic to fish except at extremely high levels, and can be considered harmless. Nitrate is used by plants, including algae, for food. This constant change from ammonia to nitrite to nitrate is called the nitrogen cycle (Figure 1).
In ponds this process takes place in the surface layers of the mud, but in tanks or aquaria, a special place has to be provided for the bacteria to live and flourish. This is called a biological filter, or biofilter. For further information on biofilters, see Florida Cooperative Service Extension publication FA-12, Principles of Water Recirculation and Filtration in Aquaculture.
One important point to mention about the nitrogen cycle, is that both groups of nitrifying bacteria need oxygen to function. If oxygen levels are not sufficient, the process can break down, and you will start seeing levels of ammonia and nitrite rise in the system.
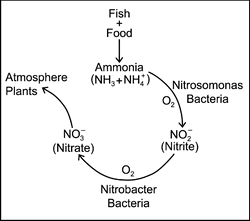 Figure 1. The nitrogen cycle. Nitrifying bacteria utilize oxygen to convert ammonia and nitrite into the nontoxic byproduct, nitrate, which is then used by plants or returned to the atmosphere. |
Ammonia Testing
Everyone involved in aquaculture should invest in a water quality test kit. A good water quality management program will result in fewer fish disease problems, better growth and less use of chemical treatments. The cost of a water quality test kit will pay for itself many times over, both in numbers of fish saved, and increased production.
Most commercial ammonia test kits measure the Total Ammonia Nitrogen (TAN). As mentioned before, it is the fraction of the TAN which is in the unionized ammonia (UIA) form which is toxic. This fraction can be determined from the TAN, if you also test the temperature and pH of the water. At high temperatures and high pH, there is more UIA. Therefore, a good ammonia test kit would include a TAN test, a pH test, and a thermometer.
When Should Ammonia Be Tested?
Ammonia should be tested every 10 to 14 days in ponds, and at least once a week in tanks. If the tanks are using a common biofilter (i.e. a recirculating system), you won't need to check every tank individually. Records should be kept for all tests, and whenever ammonia is found, the frequency of testing should be increased until the problem is corrected. Whenever fish are sick, test the water quality. Ammonia is responsible for more unexplained losses in aquaculture than any other water quality parameter.
At concentrations typically found in ponds and tanks, it is colorless and odorless, so the only way to know if it is present is to test for it. Fish submitted to a diagnostic laboratory are tested for diseases (bacteria, parasites, fungi or viruses) only. It is the producers responsibility to test the water quality, which can easily be the underlying problem.
Interpreting the Ammonia Test
In healthy ponds and tanks, ammonia levels should always be ZERO. Presence of ammonia is an indication that something in the system is out of balance. Therefore, ANY ammonia in a pond or tank should alert the producer to start corrective measures. However, it is only the unionized ammonia (UIA) which is toxic to fish. This toxicity begins as low as 0.05 mg/l, so a positive TAN test needs to be followed by a test to find the actual concentration of UIA.
To do this, the temperature and pH need to be measured. Once the pH and temperature are known, one can determine the fraction of UIA present from Table 1. Find the temperature on the top, and the pH on the left. The number taken from the table is multiplied by the TAN to give the UIA in mg/l (ppm). This calculation is summarized in Figure 2 and an example is given in Figure 3. Anytime that the UIA is higher than 0.05 mg/l the fish are being damaged. As the concentration rises above 0.05 mg/l it causes more and more damage and at 2.0 mg/l fish will die.
Again, ANY ammonia is indicative of a problem in the system and corrective measures should be taken by the producer.
 Figure 2. Step-by-step buide to calculating unionized (toxic) ammonia. UIA of 0.05 mg/l may harm fish. As UIA approaches 2.0 mg/l fish will begin to die. |
 Figure 3. An example of how to calculate UIA using the factors in Table 1. |
Management of an Ammonia Problem
Since the major source of ammonia is fish feed, the first thing to do when ammonia is present is to reduce or eliminate feeding. Fish are not likely to eat during periods of ammonia stress and the uneaten feed will only make the situation worse. Overfeeding is a major cause of ammonia problems, and stopping the feeding will allow the natural nitrogen cycle to "catch up" with the nutrient load. If at all possible, a 25% to 50% water change will help to remove some of the ammonia. This is only feasible in small ponds or tanks, so don't try to solve an ammonia problem in a large pond by this method.
In ponds, phosphate fertilizer may help to relieve high TAN levels over a period of days by stimulating plant growth which helps remove ammonia from the system. Use a 0-20-0 fertilizer at a rate of 40 pounds per acre. This may not help fast enough in an acute ammonia crisis. If phosphate is not limited in the pond, this method will not be effective at all. In tanks without a biofilter, the producer should consider incorporating one.
Summary
Ammonia is a colorless, odorless substance which can accumulate in aquaculture systems and cause direct mortalities, a decrease in production, and increased incidence of many diseases. The only way to detect its presence is by testing for it, and a fish farmer should not only invest in a water quality test kit, but learn to use it on a regular basis.
Commercial test kits only measure the Total Ammonia Nitrogen (TAN). When this test is positive, producers can determine the fraction of toxic unionized ammonia (UIA) if the pH and temperature are also measured. This information is found in Table 1, and an example calculation is found in Figure 3.
When ammonia is present, food should not be added to the system until the problem is corrected. In small systems, a water change will help, and in large ponds, 0-20-0 fertilizer may help.
By testing for ammonia on a regular basis, most ammonia problems can be minimized, since corrective measures can be taken before fish start to die. Once fish have started to die, it is difficult to correct an ammonia problem without losing more fish.
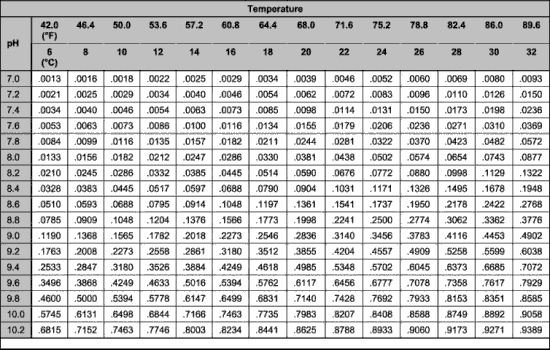 Table 1. Fraction of unionized ammonia in aqueous solution at different pH values and temperatures. Calculated from data in Emmerson et al. (1975). To calculate the amount of unionized ammonia present, the Total Ammonia Nitrogen (TAN) must be multiplied by the appropriate factor selected from this table using the pH and temperature from your water sample. See the example in Figure 3. |

Reviewed February 2006

