Several known species of Perkinsus can cause disease in shellfish. However, it is not always clear which species of shellfish will become infected or clinically diseased by which species of Perkinsus. It seems that Perkinsus-like organisms may be an incidental finding in some instances, and in other instances, the Perkinsus-like organisms appear to cause disease. There are most likely species of Perkinsus that have not yet been described.
Detection of these organisms in an infected animal may be possible during a routine necropsy; however, light infections may be difficult to diagnose. When Perkinsus is found, determining which species is present requires specialized molecular testing that few labs are able to do. Because of the potential importance of these organisms in shellfish disease, it is strongly recommended that an expert in shellfish diseases be consulted when Perkinsus is found or suspected.
Of the Perkinsus species, P. marinus and P. olseni can have a severe impact on bivalve molluscs and consequently are economically important parasites. This document primarily addresses these two species. Table 1 lists the known species of Perkinsus, the known hosts, geographical location where they have been found, and reportability.
Signs of Perkinsus infection in molluscs
Clinical signs of disease in infected molluscs are non-specific. Infected animals may gape, have a pale digestive gland, lose body condition, demonstrate mantle retraction, and contain white nodules in various tissues. Or, they may be found dead.
Infection with either P. marinus or P. olseni is frequently lethal in susceptible molluscs, though an infected animal may live for several years before showing clinical signs of disease or abruptly dying.
Perkinsus marinus
Perkinsus marinus, the cause of Dermo disease in oysters, is found on the East Coast of the USA from Maine to Florida, and along the Gulf of Mexico Coast to the Yucatan Peninsula, and the Pacific Coast of Mexico. The recent northward range extension of P. marinus into Delaware Bay, New Jersey, Cape Cod and Maine, USA, is attributed to repeated introductions over several years in conjunction with recent increases in winter sea-surface temperatures.
Perkinsus marinus causes disease and mortality in the Eastern oyster Crassostrea virginica, a commonly cultured and wild-harvested food species. In some areas, up to 100% of C. virginica may be infected, and the resulting disease is usually fatal. The presence of this parasite can damage commercial fisheries and severely limits commercial production of the Eastern oyster.
The Pacific oyster C. gigas, the Asian oyster C. ariakensis, and the Mangrove oyster C. rhizophorae, as well as other oysters, can be infected with P. marinus but do not show signs of disease except under experimental conditions.
The hard clam Mercenaria mercenaria, another commonly cultured species, is not susceptible to infection with P. marinus. However, if hard clams are located near infected oysters, they may ingest P. marinus and act as a mechanical vector. Subsequent movement of these clams may introduce a different strain of P. marinus to a susceptible population of oysters.
Perkinsus olseni
Perkinsus olseni is a protistan parasite of many molluscs and was first identified in several species of the gastropod abalone Haliotis along the coast of Australia. Venerid clams are particularly susceptible to infection with P. olseni. Venerid clams are found around the world and include species such as the hard clam Mercenaria mercenaria, and the Manila clam Venerupis philippinarum. P. olseni is also associated with high mortalities of many bivalve molluscs, including the giant clams Tridacna spp. that are frequently kept in tropical reef aquaria.
Perkinsus olseni is primarily found throughout the Pacific Ocean, including Korea, Australia, New Zealand, Japan, China, Portugal, Spain, Italy, France, Viet Nam, Thailand, and Uruguay. To date, it has not been shown to cause disease in the hard clam M. mercenaria or the Eastern oyster C. virginica in North America.
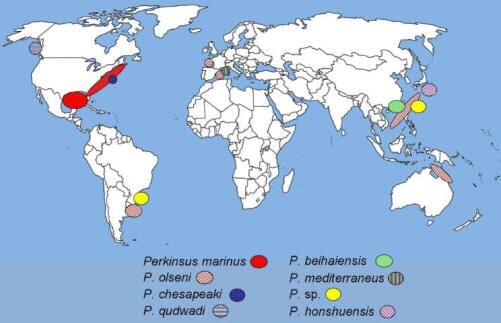
Other Perkinsus species of North American bivalves
The soft shell clam Mya arenaria and Baltic clam Macoma balthica, found in Virginia and Maryland, are susceptible to infection with Perkinsus chesapeaki, and a Perkinsus species has been identified in the hard clam M. mercenaria. These two species of Perkinsus rarely cause severe disease or kill their host bivalves.
The life cycle of Perkinsus species All Perkinsus species have a similar life cycle consisting of multiple stages, all of which cause tissue damage in the mollusc and are infective.
The first stage is the zoospore. The zoospore is motile, which means that it can swim freely in the water by moving its two flagella. The host ingests the zoospore, which then becomes a trophozoite. The non-motile trophozoite multiplies in a vegetative form within the host tissue. Perkinsus marinus primarily infects hemocytes (the blood cells of the mollusc). In the infected tissue, trophozoites develop a large vacuole and a displaced nucleus. The infected cell is similar in appearance to a signet ring, and is called a signet ring cell. Mature trophozoites divide by successive binary fission resulting in the release of 8–16 immature trophozoites. These immature trophozoites may continue to infect the remaining tissues of the host, or they may be released into the water in the feces or when the host dies. As the infection spreads within the mollusc, the host tissues are severely damaged. In seawater, the trophozoite greatly enlarges, and goes through several developmental stages, culminating in the release of zoospores. The new zoospores use their flagella to swim to new hosts, and the cycle repeats.
Transmission of Perkinsus species
The parasite is spread directly from animal to animal and when susceptible animals ingest water containing Perkinsus. The degeneration of a dead infected mollusc releases many infective parasites. In contrast, live infected molluscs shed low numbers of parasites in their feces and pseudofeces. Infected molluscs should never be moved to an area with uninfected molluscs.
Proliferation of all Perkinsus species is correlated with warm summer water temperatures when pathogenicity and associated mortalities are highest. Water temperatures over 25°C (77°F) result in rapid increase in propagation of P. marinus. In animals infected with P. olseni, infection peaks when water temperatures are 15°C or warmer (59°F or warmer), and declines when water temperatures reach 10°C or cooler (50°F or cooler).
Salinity also affects both the intensity and incidence of Perkinsus infections. Though P. marinus can survive in host tissues at salinities less than 9 grams per liter (g/L), it does not multiply in the host tissue and host mortality does not occur. However, P. marinus will multiply in host tissue at a salinity range of 9–12 g/L, and infections intensify when salinity rises above 12 g/L. Little is known about the effect of salinity on P. olseni, but research suggests this parasite thrives at a salinity of 25 g/L, and does not tolerate salinities of less than 15 g/L.
Consequently, high mortalities of P. marinus-infected oysters are commonly seen in the late summer months and in times of drought. Researchers have observed a higher intensity of infection associated with the stressors of spawning activity in molluscs in the late summer months.
Stressful conditions such as harvest and high stocking densities may increase the susceptibility of molluscs to infection.
Diagnosis of Perkinsus species
Perkinsus infections in molluscs cannot be diagnosed without specialised testing.
Wet mount examination of hemocytes from oysters infected with P. marinus or tissues containing nodules from molluscs infected with P. olseni may reveal spherical cells with an eccentric vacuole (signet ring appearance) or mature trophozoites within hemocytes. However, the absence of these forms of the parasites on wet mount examination does not mean the animal is not infected.
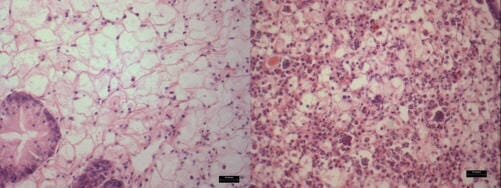
Histologic exam may reveal an inflammatory response (Figure 2) in association with the various stages of the parasite. This method is not conclusive for identifying Perkinsus infection, however, because the infected animal may have a low-intensity infection that is not easily detectable with histology.
A standard method for identification of infected molluscs is culture in Ray's fluid thioglycollate medium (FTM). After five to seven days of culture of affected host tissues in FTM, trophozoites enlarge to become hypnospores that are more easily seen after staining with Lugol's iodine.
This method is effective for diagnosis of Perkinsus infection, but it does not distinguish between the different species of Perkinsus.
Species identification requires polymerase chain reaction (PCR) testing, a molecular technique that utilizes specific DNA (gene) fragments.
Regulatory concerns
The World Organisation for Animal Health (OIE) is the international authority for improving animal health, and as such designates, or lists, animal diseases of concern. Both P. marinus and P. olseni are OIE-listed disease agents. This means any mollusc diagnosed with P. marinus or P. olseni must be reported to the appropriate authorities. In the United States, that authority is the state veterinarian and the United States Department of Agriculture Animal and Plant Health Inspection Service (USDA-APHIS) area veterinarian in charge (AVIC).
In addition, many states require testing of bivalve molluscs for Perkinsus sp. and other pathogens before they are moved across state lines. The office of the state veterinarian (a list can be found here: http://www.usaha.org/StateAnimalHealthOfficials.pdf ) or the state department of natural resources should be contacted for current regulations. A list of state requirements (current in 2002) is available in the Proceedings of the Eastern United States Interstate Shellfish Seed Transport Workshop (see References section). Laboratory, state, national, and international guidelines for testing will determine the number of animals that need to be checked.
Treatment and disinfection of facilities that may be contaminated with Perkinsus species
There is no treatment for molluscs infected with any of the Perkinsus species.
Mortalities may be decreased by correcting adverse environmental conditions and by reducing stocking density, but the animals that survive will still be infected and can carry the disease to naïve (uninfected) susceptible animals. State and/or federal authorities may require destruction of infected animals and disinfection of contaminated holding containers.
Research to develop a disease-resistant oyster has been promising, but expensive. Some New England states recommended managing infected oyster areas by removing infected oysters, but this practice has no significant impact, and it's possible that disease-resistant oysters may be removed in the process.
Perkinsus species have a thick cell wall that helps them survive adverse conditions. The length of time P. marinus can survive outside its host is unknown, but some stages of P. olseni can survive outside the host for up to 129 days at 10°C. P. olseni can survive freezing when in host tissue for as long as 197 days, so tissues from any mollusc from the geographical region where P. olseni is found should not be disposed into open seawater.
Both P. marinus and P. olseni in seawater can be killed when exposed to freshwater at room temperature for 10 minutes; freshwater exposure will not have any effect on the parasites in host tissues. Chlorine at greater than 300 milligrams per liter (mg/L) is effective against both species. Ultraviolet light sterilization greater than 28,000 microwatts second per square centimeter (µWs/cm2) is effective against P. marinus, but 60,000 µWs/cm2 is required to kill P. olseni.
Food Safety
The Perkinsus parasites are not infective to humans, thus Perkinsus-infected bivalve molluscs are safe for human consumption.
Summary Perkinsus marinus, a protistan parasite, must be considered in mortalities of oysters from the eastern coast of the United States and the coast of the Gulf of Mexico. Perkinsus olseni, a related protistan parasite, must be considered in mortalities of bivalve molluscs and gastropods (such as abalone) from the Pacific Ocean.
Perkinsus parasites do not infect humans.
Culture of mollusc tissues in Ray's fluid thioglycollate medium should be performed in moribund and fresh dead animals. If the culture results are positive, identification of the Perkinsus species should be made using a PCR-based protocol. If the animals are infected with P. marinus or P. olseni, the state veterinarian and the USDA-APHIS AVIC should be informed immediately.


