The digestive tract of fish can be described basically as a muscular tube lined by a mucous membrane of columnar epithelial cells that exhibit regional variations in structure and function (Ringo et al, 2007). It's major function is to process the ingested feed material and to digest and degrade this into a form that can be easily absorbed and assimilated by the animal and thereby supply dietary nutrients to the body tissues.
Digestion by its very nature is a complex process involving enzyme and fluid secretions, motility, absorption and ultimately evacuation. And a healthy digestive system is crucial for optimal animal performance.
The large surface area of the gastro-intestinal (GI) tract is necessary for optimum nutrient absorption and also to allow the efficient passive transfer of nutrients into the bloodstream. However, the gut is protected by only a single layer of epithelial cells and damage to these structures can therefore impact nutrient uptake and affect growth and performance.
Integrity
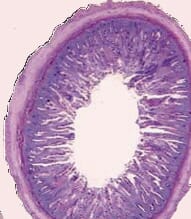 |
| Figure 1: Rainbow trout gut transect showing the more complex villi structures in the Bio-Mos fed fish Top: Bio-Mos Bottom: control |
The histological study of the intestine is important in establishing the status of structural integrity. It acts as a tool that helps improve our understanding of dietary influences that can either positively improve structure or factors such as infectious diseases or anti-nutritional components that may ultimately cause physical damage.
The GI tract is a major route of infection in fish and an array of protection systems exist within it to limit the risk of damage to the GI tract. Mucosa is critical in digestion, absorption and metabolic processes and acts as a barrier to pathogenic infections preventing both viable and non-viable bacteria and their bacterial products from migrating from the intestinal lumen through the epithileal mucosa to infect otherwise sterile tissues.
Mucosa plays a role in the electrolyte balance, immune response and endocrine functions. Mucins and glycoproteins associated with the intestinal brush border serve as important barriers protecting the absorptive surface from feedstuffs, bacteria colonization and toxins. In addition, endogenous acids, digestive enzymes and bile reduce bacterial growth, while digestive flow and peristaltic movements transport the foods through the tract limiting bacterial development.
Farmed fish are vulnerable to ubiquitous opportunistic bacterial pathogens - often striking when fish stocks are stressed.
Stress and environmental conditions are closely interlinked and one of the consequences of stress can be the loss of intestinal integrity causing enhanced epithelial permeability that may lead to enhanced uptake of macromolecules, bacterial products and antigens across the epithelium. This can lead to reduced performance and susceptibility to disease. Therefore the interactions between intestinal microflora, gut morphology, the immune system and nutrient uptake will have a major influence on the animals health and performance.
Enteritis and poor gut morphology can lead to inefficient feed conversion and the repair of damaged enterocytes is an energy consuming activity which in turn directs valuable resources from growth to the more immediate urgency of tissue repair and maintenance.
MOS Benefits
Mannan oligosaccharides (MOS) have been shown to affect gut health by pathogen adsorption and immune modulation. However the different fermentation conditions, the different strains of yeast used and the different processing methods used in their manufacture can alter their function. (Newman, 2007).
Bio-Mos® is a MOS derived from the outer cell of a specific strain of yeast Saccharomyces cerevisiae 1062. Its use in terrestrial animals has been well documented, but it has is only recently been shown to offer advantages to farmed fish.
One of the key benefits of Bio-Mos is its ability to bind or agglutinate bacteria, which prevents it colonising in the gut and so causing infection. Table 1 shows the agglutination of Bio-Mos to a number of strains of bacteria known to cause disease in fish and shrimp.
| Table 1: Results of agglutination tests using Bio-Mos | |
| Bacterial strain | Aggregation to Bio-Mos |
|---|---|
| Aeromonas veroni #9071 | Positive |
| A. veroni field isolate | Positive |
| A. hydrophilia #35654 | Positive |
| A. hydrophilia #35654 | Positive |
| A. caviae #15908 | Positive |
| A. caviae field isolate | Negative |
| Edwardsiella tards isolate 1 | Positive |
| Edwardsiella tarda fiels isolate | Negative |
| V. fluvialis field isolate | Positive |
| V. parahemolyticus CSUH | Positive |
| V. (Listonella) anguillarum | Positive |
| V. vulnificus field isolate | Positive |
| Photobacterium (Pasteurella) damselae ssp Piscidia #17911 | Positive |
Trial results obtained with Bio-Mos Microbiology by Zhou and Li (2004) reported that the intestinal microbial populations in Jian carp, were significantly affected by the dietary inclusion of 0.24 percent of Bio-Mos. The presence of Escherichia coli in the intestinal digesta were significantly decreased while significant increases were observed in bifidobacterium and lactobacillus.
Recent work by Dimtroglou et al (2007) demonstrated that Bio-Mos significantly reduced the bacterial load in the gut of both rainbow trout and sea bream by reducing the total aerobically cultivated bacteria. In the case of sea bream, the percentage contribution of the various aerobically cultivated bacterial families to the total count was not significantly altered between the Bio-Mos fed fish and the controls. However, in the case of rainbow trout, the Bio-Mos fed fish had reduced numbers of Micrococcus spp., Staphylococcus spp., Aeromonas/ Vibrio spp and other unidentified Gram + bacteria. Increases were also seen in Acinetobacteria spp., Pseudomonads spp. and Enterobacteria spp.
Conclusions were that the Bio-Mos promoted the colonisation of beneficial bacteria - those associated with the natural gut flora of the rainbow trout.
The differences observed between the two species were reported as being due to:
- the fact that marine fish effectively ‘drink’ significant amounts of water, to maintain osmotic regulation, via their gastrointestinal tract
- the culture conditions - the rainbow trout were reared in an open flow system while the sea bream were reared in a closed circuit system.
Morphology Effects
The effect of Bio-Mos on the gastrointestinal morphology of several species: rainbow trout, salmon, sole and sea bream has been reported recently (Dimitroglou et al, 2007).
In adult rainbow trout and sole, the MOS was found to have a significant effect on the external and internal perimeter ratio both in the anterior and in the posterior gut regions, indicating a more complicated architectural gut structure with longer villi, and hence a large surface area for nutrient absorption (See Figure 1).
The inclusion of the MOS improved the microvilli density only in the posterior gut region of the fresh water fish, rainbow trout. In marine fish species, salmon, sole and sea bream, Bio-Mos produced a more pronounced integral effect by increasing the microvilli density in both the anterior and posterior gut regions. The microvilli length was also significantly increased in all the adult fish in both the anterior and posterior gut regions. In juvenile rainbow trout the effect of Bio-Mos on the microvilli length was restricted to the anterior part of the gut.
Observations of microvilli structures of salmon,showed that microvilli density of the Bio-Mos fed groups of fish was 4.62 times greater than that of the control groups. Microvilli length was also 21.1 per cent higher in the in the Bio-Mos fed groups with less detected damage.
Clear Advantages
It is clear from these results that feeding a MOS can have a significant effect on gut morphology. The changes observed, including increased perimeter ratio, increased villi and microvilli density and length, indicate that the absorptive surface of the gut is improved, and so a better absorptive capability appears to be possible. Increased microvilli density without signs of damage or irritation will also eventually lead to an increase of the absorptive capability of the enterocytes.
In the case of sole, the Bio-Mos was added to the feed in order to improve the condition of vaccinated and non-vaccinated fish. In both cases the MOS improved the villi structure and increased the microvilli density. A disease outbreak in these fish, and the overall mortalities, were reduced in the MOS fed groups when compared to the control groups. (Dimitroglou et al, 2006).
Growth, performance and immune parameters
In addition to the improvements seen in gut morphology and function, using a MOS has been shown to improve growth, performance and immune status in a number of fish and crustacean species. Zhou and Li (2004), in addition to the changes in gut microflora mentioned previously, showed higher weight gains, improved feed conversion ratio (FCR) and immune parameters in Jian carp when Bio-Mos was included in the diet.
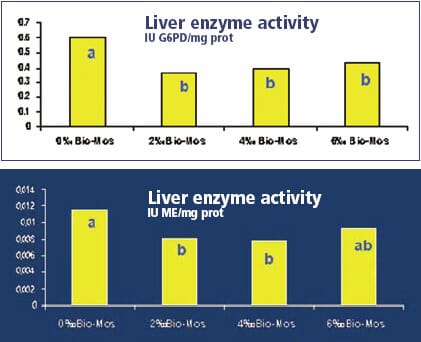
Similar effects of Bio-Mos fed carp have also been reported (I), rainbow trout (I)and catfish (Bogut et al, 2006).
Investigations by Daniels in 2005 also reported decreased mortalities and improved survival rates in lobster larvae fed an MOS in to stage IV of production. These improvements in performance have also been observed for marine fish species.
Disease resistance to bacterial infection, both by cohabitative challenge and by direct inoculation in the gut, has also shown to be better when Bio-Mos was incorporated in the diets. It has resulted in improve liver health and function which may well be an indicator of better utilisation of dietary nutrients (as shown in the above graphs). This in association with improved gut morphology may suggest reasons for better growth and performance.
Conclusion
Using a MOS, (and in this case Bio-Mos) has been shown to be an effective tool for increasing performance and health status of a number of important commercial fish species.
Acting in a prophylactic manner it provides multiple benefits when incorporated in to aqua feed diets by not only improving the GI morphology but also its function through an increased absorptive surface and better absorptive capability. It also interacts with the immune system in a modulatory manner and alters enzyme activity promoting the better utilisation of dietary nutrients thereby improving performance characteristics and immune function.
References
These are available in PDF version below.Further Reading
|
|
- To read the entire report click here. |
* Authors:
John Sweetman - Alltech Aqua, Cephalonia, Greece.
Arkadios Dimitroglou - Fish Nutrition and Aquaculture Research Group, University of Plymouth, UK
Simon Davies - Fish Nutrition and Aquaculture Research Group, University of Plymouth, UK
Silvia Torrecillas - Grupo de Investigación en Acuicultura (GIA). ICCM & IUSA, Las Palmas de Gran Canaria, Canary Islands, Spain.
This paper was first published in International Aquafeed, January-February 2008. Reproduced courtesy of Alltech Inc.

