Three species are known to occur in UK freshwaters: Argulus foliaceus, Argulus coregoni and Argulus japonicus. Two of these species, A. foliaceus and A. coregoni, are regarded as native to the UK (Fig. 1). These species are easily discriminated since adult A. coregoni are over twice the size of adult A. foliaceus and have pointed, rather than rounded, abdominal lobes. A. japonicus is thought to be non-native and introduced from Asia through the ornamental trade. This species looks very similar to A. foliaceus and can only be discriminated at the microscopic level.
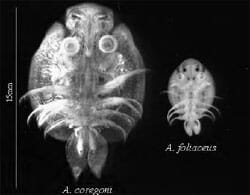 |
| Figure 1. Ventral view of adult A. coregoni and A. foliaceus. |
A. foliaceus is widely recorded throughout the world, typically inhabiting nutrient rich lakes, although it can tolerate salinities of up to 8-12ppt at temperatures up to 25°C. A. coregoni typically infects fish in rivers, streams and cool oligotrophic lakes with a large flow. A. japonicus is recorded throughout Europe, Africa, North America and Asia. At present there are limited records of A. japonicus in the UK, and due to difficulty in differentiating the two species it is possible that in some cases it has been misidentified as A. foliaceus.
Many of the early records of Argulus spp. in Europe refer to them as a problem in carp farming, with much of the German literature referring to the ‘Carp Louse’. In recent years however, Argulus spp. have been reported to cause problems in UK stillwater trout fisheries. A survey of such fisheries found 29% of them suffered from problem infections by the parasite in the year 2000. A. foliaceus was responsible for all but one of the observed infections, the other being caused by A. coregoni.
Infected fish can suffer significant reductions in condition factor, reducing the aesthetic appeal to anglers of fish in a sports fishery. Heavy infections may also cause noticeable behavioural changes, which develop as the severity of the outbreak increases. In the early stages of an infection, fish in lakes are reported to jump, flash and swim erratically, possibly in an attempt to rid themselves of lice. Following this, feeding is reduced and the fish lose condition and become difficult to catch by angling. As infection levels rise, fish have been observed to swim in tight shoals, and in serious cases substantial mortality can occur.
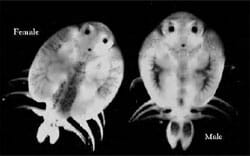 |
| Figure 2. Dorsal view of adult female and male A. foliaceus. |
Argulus spp. reproduce sexually and male and female parasites can mate on or off the host. The adult female has a single median ovary, which runs the entire length of the body. Although several matings may be observed, it is thought that one is sufficient to fertilise all the eggs that a female will ever produce. Adult males and females are easily distinguished as males have large dark spots on each of the abdominal lobes, and females have spotted pigmentation running up the centre of the dorsal surface of the carapace, covering the ovary (Fig. 2).
Argulus spp. in the UK lay their eggs off the host on a suitable hard surface, in rows that are attached by a gelatinous material that hardens on contact with water. Eggs of A. foliaceus and A. japonicus are laid in batches comprising 2-4 rows of up to 400 eggs, whereas A. coregoni lays its eggs in mats containing up to 900 eggs. The parasite can lay up to 10 batches of eggs, but most only lay once. Individual eggs are ovoid, and measure approximately 0.2 mm x 0.3 mm. On laying, the eggs are white to pale yellow in colour, but within 24 hours their colour changes to a deeper yellow / light brown. Eggs are firmly adhered to their attachment surface and, unlike snail eggs, are not coated in jelly. The parasite hatches as a stage known as a metanauplius, which is between 0.6 and 0.8mm in length (Fig. 3).
 |
| Figure 3. A. foliaceus metanauplius hatching from egg string. |
Hatching times are dependant on temperature, becoming shorter as temperature increases. Below 8-10°C, eggs of the three UK species do not appear to develop, which is possibly a mechanism to increase survival of the metanauplius by ensuring hatching only occurs in the more favourable conditions of spring and summer. Eggs that over-winter have a much reduced hatching success, but can survive at low temperature for up to 2 years.
In the UK, numbers of argulids on fish tend to be low in winter, with laboratory experiments suggesting that most lice die in cold conditions and after periods of reproduction. A rise in temperature in spring causes eggs to start hatching. These early generations give rise to subsequent generations that cause the population number to peak towards the end of summer/start of autumn. As the temperature falls with the advent of winter recruitment stops and the population decreases once again. Any adult lice surviving the winter become relatively inactive until the water temperature increases above 10°C, when they will leave their host to lay eggs.
Studies have shown that in trout fisheries, low water clarity, slow rates of stock turnover (the rate at which the lakes standing stock of trout is removed and replaced) and high temperatures show a significant correlation with high numbers of A. foliaceus on trout. Low water clarity was also associated with reduced stock turnover, possibly as the reduced clarity changes the distance over which trout are likely to react to prey items, thus reducing the likelihood of being caught. These results suggest that high numbers of A. foliaceus alone may not affect the catch rates unless working in combination with other environmental factors. The mechanisms behind the effects of water clarity and stock turnover on the parasites dynamics are currently unknown, although several hypotheses have been developed. It is likely that fast stock turnovers reduce the parasite numbers through removing infected fish before the parasite can leave its host to lay the eggs required to produce subsequent generations. Low water clarity may influence the parasites dynamics in several ways. Firstly through its effect of slowing stock turnover, secondly through a reduction in predation on the parasite by trout (which readily eat them in the laboratory), and finally reduced water clarity may change host behaviour in a way that would increase infection success. Further research is being conducted between Cefas and the Institute of Aquaculture (University of Stirling) in order to determine the mechanisms driving these relationships and to develop targeted management strategies.
Winter/Spring 2007

