Introduction
The first International Symposium on the Reproductive Physiology of Fish was held 30 years ago, in September 1977, at the Biological Research Station of Paimpont, in Brittany. It was the result of a pioneer initiative by Roland Billard, head of a research group on fish reproductive physiology, which was founded a few years ago in Jouy-en-Josas (France). The symposium gathered approximately 80 participants, and oral presentations were published in a special issue of the Annales de biologie animale, biochimie, biophysique, Vol. 18(4). Most of them were basic studies concerning mainly gonadotropins and gametogenesis, with a few devoted to direct applications for aquaculture (Tab. I).
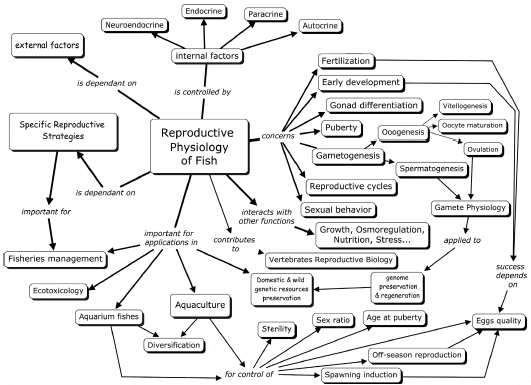
Reproductive physiology of finfish (RPF) is a vast scientific field, which directly concerns a set of physiological processes essential for reproduction, beginning with egg fertilization and ending with sexual behaviour and spawning. Processes include gonad differentiation, puberty, male and female gametogenesis, and the timing of reproductive cycles (Fig. 1), all regulated by numerous neuroendocrine, endocrine, paracrine and autocrine factors, all along the brainpituitary- gonadal (BPG) axis. These processes also interact with other important physiological functions such as growth (Le Gac et al., 1993), nutrition (Izquierdo et al., 2001), osmoregulation (Haffray et al., 1995; Le Francois and Blier, 2003), and response to stress factors (Schreck et al., 2001)… RPF is also highly dependent on external factors such as temperature, photoperiod and water salinity, which may modify the various stages of the reproductive process, depending on the species. In fact, fish exhibit a huge variety of specific reproductive strategies and tactics (Wooton, 1984), with just as many specific adaptations in terms of physiological regulation.
It is now well established that scientific knowledge in the RPF field has numerous applications. This is obviously the case for aquaculture production of many species reared for human consumption, with applications such as hormonal spawning induction (Billard, 1993; Zohar and Mylonas, 2001), off-season spawning (Bromage et al., 2001), broodstock and gamete management (Billard, 1992; Bromage et al., 1992), sex ratio control (Baroiller et al., 1999), and age at puberty (Okuzawa, 2002). The same kind of innovations might also be useful for the production of aquarium pet species, thus reducing collection pressure in the wild. In both cases, diversification of species is generally sought thus justifying the development of comparative research on reproductive strategies and physiological regulations. Preservation of domesticated and wild genetic resources, through gamete storage and/or embryo regeneration, is another research area (gamete physiology) with current and potential applications… Fisheries management might also take advantage of knowledge concerning the reproductive potential of species in order to improve modelling of exploited populations. Finally, the recognition of the sensitivity of fish reproductive processes to the presence of xenobiotics in the aquatic environment (Jalabert et al., 2000) has been widely utilized in the development of a variety of endocrine disruption biomarkers (Hutchinson et al., 2006). From a more general point of view, it appears that finfish are appropriate models not only from the perspective of ecotoxicological risks, but also in terms of comparative biology, due to the significant similarity of basic aspects of the BPG axis among vertebrates (Blazquez et al., 1998).
| Table I. - Number of articles (in brackets) published in Annales de biologie animale, biochimie, biophysique, Vol. 18(4) after the Paimpont symposium; reports on specified species or groups of species, or concerning basic or applied scientific fields (each article may be counted in several categories). | ||||||
| Species or group of species | Salmonids (27), Cyprinids (6), European eel (3), Sparidae (3), Mugilidae (2), sea bass (2), Dogfish (1) | |||||
|---|---|---|---|---|---|---|
| Basis scientific fields | Pituitary gonadotropins (biochemistry, cellular sources, secretion, neuroendocrine control, steroid feed-back (24), gametogenesis (oogenesis (15), spermatogenesis (3)), sex steroids (7), temperature and photoperiod (4), reproductive cycles (4), sex differentiation (2), sexual behaviour (1) | |||||
| Applications of aquaculture | Hormonal spawning induction (5), sperm conservation and quality (4), off-season spawning (3), sterilization (1) | |||||
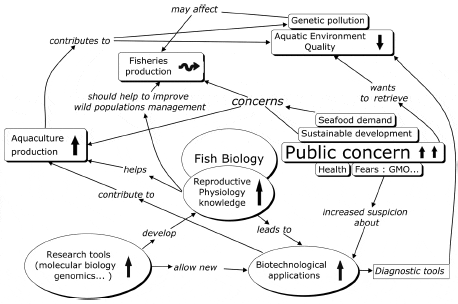
Development of research in the RPF field during the last three decades cannot be dissociated from the concomitant evolution of the general socio-economic context (Fig. 2). According to FAO data, the world finfish aquaculture production has grown from around 3.6% of total finfish production in 1975 (58.4 million tons), to 27.6% of total production (about 110 million tons) in 2005. In addition, since capture fisheries have been stagnating for the last 15 years, the world fisheries production increase during this period is due entirely to aquaculture production development. This increase is driven by both the increasing world demand for fish (which capture fisheries can no longer meet due to overexploitation of natural resources), and by the application of new biotechnology in all fields of fish biology, in particular reproductive biology. An increase in public concern has also been observed during this period, driven by fears about the possible negative health and environmental impacts of new production technologies, and expressed through a growing demand for sustainable production methods.
Since the publication of an exhaustive review of knowledge in the field of RPF 26 years ago (Sundararaj, 1981), the number of scientific papers published each year in the field has increased tremendously. Therefore, the present article does not seek to be exhaustive, but will focus on a few significant discoveries in selected areas of RPF: gonadotropins and gonadotropin releasing hormones (GnRH) as key regulators of reproductive function; and puberty as an example of a reproductive function process dependant on specific reproductive strategies, exhibiting various reproductive tactics and regulated by a cascade of endocrine, paracrine and autocrine factors. Finally, some examples of economically relevant applications of RPF knowledge will be recalled.
Fish gonadotropins: from one GtH in 1977… to FSH and LH today
In 1977, relatively purified gonadotropin (GtH) preparations had been extracted from carp, salmon, sturgeon and trout pituitaries through biochemical separation techniques. Some of them were already sufficiently homogeneous for the purposes of radio-immuno assays in endocrinological studies. However, even after validation of purification steps by global biossays typical of final gametogenesis steps, such as spermiation or intrafollicular oocyte maturation (Breton et al., 1976), there was still no clear biochemical or biological evidence of gonadotropin duality in fish. Besides, cytological observations of pituitary cells had suggested the presence of one or two cell types in teleost fish, depending on the species. Yet, the first isolation of two types of gonadotropins, exhibiting predominant vitellogenic or maturational activities, was achieved in the plaice (Ng and Idler, 1978). Later, two distinct salmonid GtH, GtH I and GtH II, were isolated and characterized by their respective steroidogenic potency in vitro (Suzuki et al., 1988; Swanson et al., 1991). Since then, both GtHs have been prepared in many other species, through classical purification methods and/or, more frequently, sequencing and homology cDNA cloning: common carp, grass carp, eel, killifish, striped sea-bass, channel catfish, European sea-bass, red sea-bream, tilapia, Atlantic halibut, grouper (Li et al., 2005; Yaron et al., 2003). Although the structural homology of the β subunit of a purified GtH from chinook salmon to human LH was already suggested twenty years ago by partial protein sequencing and cDNA cloning (Trinh et al., 1986), the identification of GtH I and GtH II to FSH and LH respectively was finally formally adopted at the 6th ISRPF in Bergen in 1999. The characterization and availability of both gonadotropins have allowed specific assays to be set up and experiments at endocrinological, cellular and molecular levels to be carried out, thus leading to significant progress in the understanding of their respective physiological targets and mechanisms of action.
Regulation of pituitary gonadadotropin secretion and multiplicity of GnRH functions
Most data in the field were obtained either at the time of oocyte maturation and/or by using radio-immuno assays of GtH I (LH), the first gonadotropin available in goldfish and salmonids. Therefore, the terms “gonadotropin” or GtH in the following paragraph will refer implicitly to LH.
In 1977, the presence in the fish hypothalamus of a LHRH- like factor able to promote gonadotropin release had already been shown a few years ago (Breton et al., 1971). Mammalian synthetic LH-RH could therefore be used to observe variations of GtH secretion response in vivo, dependent on the gametogenetic stage (Weil et al., 1978). In fact, the 1st fish GnRH, sGnRH was characterized only a few years later, after purification from chum salmon brain (Sherwood et al., 1983). In the meantime, the inhibitory effects of dopamine on gonadotropin release, induced by injections of a luteinizing hormone-releasing hormone analogue (LHRHa), was demonstrated in female goldfish (Chang and Peter, 1983). From that time on, the model of gonadotropin secretion regulation was progressively enriched by numerous data. First, it had to take into account that GnRH was not only originating from the hypothalamus but also from other brain areas (Kah et al., 1993). Moreover various brain neurotransmitters, together with steroid feed-backs, appeared able to modulate the main stimulating/inhibitory roles of GnRH/dopamine, thus relaying the action of external factors. A general model of regulation was thus published (Peter and Yu, 1997), in which GnRH is identified as the main stimulatory factor in all species investigated, whereas the inhibitory role of dopamine is more or less important, depending on the species.
In addition to the control of gonadotropin release by the pituitary, GnRH has also been involved in the control of GH release in the goldfish pituitary (Marchant et al., 1989). More recently, due to the accumulation of new data favoured by the availability of molecular tools, it emerged that GnRH exhibits a large functional diversity, not only in fish, thus again boosting new comparative research in all vertebrates (Lethimonier et al., 2004; Sherwood and Adams, 2005). Data in fish concern: the multiplicity of natural variants (at least 8 variants among fish species investigated so far, with 2 to 3 variants in each species); the multiplicity of receptors (GnRHRs), each one exhibiting different affinities to different GnRH natural variants or analogues; the multiplicity of sites of expression (not only in the brain, but also in the gonads, gills, liver, kidney and other tissues, depending on the species); and the multiplicity of biological effects, such as cell proliferation and steroidogenesis in males and meiosis reinitiation in females. At the pituitary level, the development of a novel single cell real-time quantitative PCR technique has shown that multiple combinations of three GnRH receptor types could be differentially expressed in individual gonadotropic and non gonadotropic cells of tilapia Oreochromis niloticus, thus demonstrating cellular and functional heterogeneity in correlation with reproductive state (Parhar, 2005). All the above data, together with comparative data in other animals, suggest that pleiotropic GnRH function in fish might result from evolution of an ancestral gonadotropic function in primitive organisms (Kah, 2004).
Puberty in fish: an example of a reproductive trait important for aquaculture production, exhibiting a variety of specific strategies and tactics, modulated by external factors, and under complex physiological control
Puberty designates both the transition period and the underlying physiological process corresponding to the acquisition of the capacity to reproduce. In fish, puberty is generally considered as beginning with the apparition of the first spermatocytes in the male, and the initiation of vitellogenesis in the female (Le Bail, 1986), although the second criteria is controversial. Puberty is one of the main traits representative of specific reproductive strategies and tactics as defined by Wootton (1984): each species exhibits a general strategy regarding the age at puberty, such as late puberty in sturgeons or early puberty in tilapias. Depending on various environmental factors, most species are also able to exhibit adaptive tactics within this general strategy. For example, this is demonstrated by the case of precocious puberty in the sturgeon under artificial farming conditions (Doroshov et al., 1997), by precocious male puberty in salmonids, and early puberty in tilapia induced by a protein-enriched diet Oreochromis niloticus (Gunasekera et al., 1995).
Early puberty, which is observed in most farmed species, diverts much metabolic effort towards development of the gonads, although it is often preceded or accompanied by accelerated body growth that may be a desirable trait. This paradox is an important problem in fish farming, partly resolved through food restriction or control of external factors such as temperature or photoperiod. It brings to the fore the existence of a subtle physiological equilibrium between growth and puberty, modulated by external factors.
Concerning the possible internal factors determining puberty, the “gonadostat” and “missing link” concepts (Schulz and Goos, 1999) present some heuristic interest. The “Gonadostat” refers to the negative steroid feedback upon the GnRH system in mammals before puberty. The “missing link” assumes that one or more components of the Brain- Pituitary-Gonad axis are non-functional before puberty. Information has progressively been acquired through several approaches. The stimulatory effect of exogenous steroids on the gonadotropic activity in immature male fish has been demonstrated in various species, both at the brain and/or pituitary and/or blood and gonadal levels (rainbow trout (Atteke et al., 2003; Magri et al., 1985), platyfish (Schreibman et al., 1989), African catfish (Cavaco et al., 1998), seabass (Zanuy et al., 1999), (Okuzawa, 2002). This rules out the “gonadostat” concept, according to Schulz and Goos. However, whereas this effect can be accompanied by the onset of spermatogenesis in males, no stimulation of vitellogenesis has ever been reported, suggesting that a gonadal steroid is not the only “missing link”, at least in females.
Concerning the role of pituitary hormones, many feed back experiments based on the administration of exogenous steroids have given contradictory results regarding the respective role of FSH and LH, especially in salmonids (reviewed in Okusawa, 2002). However, molecular studies at the pituitary level and endocrinological studies carried out throughout puberty suggested that FSH, and to a lesser extent GH, could be involved (preferentially to LH) in spermatogenesis and vitellogenesis initiation in prepubertal rainbow trout (Gomez et al., 1999). In addition, a consistent positive relationship between plasma IGF1, estradiol-17beta, and pituitary FSH was observed during the growth of coho salmon in spring, suggesting that these factors provide an important link between body growth and oogenesis (Campbell et al., 2006).
The possible involvement of leptin, a peptidic hormone secreted by adipocytes, in the control of puberty through regulation of gonadotropins or somatolactin secretion was recently investigated respectively in trout (Weil et al., 2003) and sea bass (Peyon et al., 2003). In trout, the absence of the response of pituitary cells from immature fish to recombinant human leptin, and the stimulation of FSH and LH release only when gametogenesis was fully engaged, suggested that leptin could not be the unique trigger for puberty. Besides, the positive response of somatolactin secreting cells from prepubertal sea bass to mouse recombinant leptin suggested that leptin might play a role in this species, through growth regulation by somatolactin.
At the brain level, the recent discovery in mammals of the key role of kisspeptin and its protein-G coupled receptor (GPR54) in the control of puberty (Messager et al., 2005) was rapidly followed by GPR54 cloning and detection in individual GnRH neurons and pituitary cells of tilapia (Parhar, 2005). Interestingly, the latest techniques allowing mRNA expression profiling at the individual cell scale also demonstrated that multiple GnRH receptors (GnRH-Rs) are co-expressed, not only in lactotropes, somatotropes, luteinizing and follicle stimulating hormone cells, but also in somatolactin, thyrotrope, corticotrope and melanotrope cells (Parhar et al., 2005). Since then, expression patterns of GPR54 mRNA in the brain of grey mullet (Nocillado et al., 2007) and cobia (Mohamed et al., 2007) were observed as being concomitant with puberty onset.
In conclusion, new endocrinological and molecular evidence confirms the close inter-relations between puberty and growth regulation at all levels of the BPG axis. However, the ultimate trigger mechanism for the initiation of puberty remains unclear. Kisspeptin certainly appears to be a key part of that mechanism, but what controls its release and how? Recent research often uses a “candidate gene approach”. However, the present availability of large-scale, and high-throughput methods, in the functional genomics of whole tissue transcriptome provide hope for new discoveries. For example, it was recently observed that 15% of the 3000 genes surveyed in brain transcription profiles of early maturing Atlantic salmon “sneakers” males, immature males and immature females exhibited differential expression (Aubin-Horth et al., 2005). Pertinent candidate genes for puberty onset might be expected among these.
Concluding remarks concerning the application of fish reproductive physiology research to aquaculture
Research in RPF represents a large scientific community all over the world, sustained by public, economic and environmental concerns in many countries. Scientific approaches concern various complementary levels, from basic knowledge to directly applied research.
In many cases, practical demand, originating mainly from the fish farming sector, has been pushing research to find solutions for the control of reproductive traits of economic interest, almost independently of research into underlying physiological regulations. This has been the case concerning manipulation of external factors, such as photoperiod or temperature. Thus, photoperiod manipulations have been applied in many farmed species to control out-of-season spawning (Bromage et al., 2001) and puberty (Okuzawa, 2002). In both cases, however, precise protocols depend not only on the species but also on the genetic strain: in rainbow trout for example, the same photoperiod regime cannot be applied to an autumn-spawning and a spring-spawning strain. Moreover, interactions with other factors are either unknown or cannot be easily taken into account for practical reasons. This is the case for temperature, for which specific requirements are not limited to the spawning period, thus impairing egg quality, as shown in Arctic char (Gillet, 1991), rainbow trout (Davies and Bromage, 2002), Atlantic salmon (King et al., 2007), or halibut (Brown et al., 2006). Besides, inadequate photoperiod manipulation by itself can also impair egg quality, as in trout (Bonnet et al., 2007) or in Eurasian perch (Migaud et al., 006). In fact, the manipulation of external factors to control any reproductive step is generally limited by the ignorance of the precise mechanisms by which these factors act, and interact, on underlying physiological processes. As already underlined in the case of puberty, much can be expected from largescale and high-throughput methods applied to functional genomics of whole tissue transcriptome and proteome, to understand multiple interactions between external factors and physiological regulations.
Applications of RPF knowledge generally bring progressive improvements to aquaculture. This is the case, for example, of hormonal spawning induction. Initially a solution was provided in the form of injections of crude pituitary extracts, which were successively replaced by partially purified gonadotropin, then by various GnRH analogs, sometimes in combination with anti-dopaminergic compounds (reviewed by Zohar and Mylonas, 2001). However, rapid industrial development of aquaculture may follow on from the development of techniques that unlock a reproductive block, as in Pangasius fish farming in Vietnam. The annual farmed production of this species, based on fry capture in the wild, was stagnating at around 12500 tons until 1995. Then, the demonstration that successful maturation and ovulation could be artificially induced by serial hCG injections (Cacot et al., 2002; Cacot and Lazard, 2004), boosted annual production up to 440000 tons in 2005 and probably near 1 million tons in 2007. Most of the increase concerns Pangasius hypophthalmus species, initially considered as less valuable with regards to flesh quality. This apparent paradox results from higher fertility and easier induction treatment of Pangasius hypophthalmus, but it also points to the essential limiting factors of species domestication, such as fertility (number of eggs per fish) and practicability of possible treatments in field conditions.
Finfish aquaculture based on seed capture in the wild, or even on the capture of adults, still concerns species such as eels, tunas, groupers and yellowtails, some of which are now considered as endangered species. Therefore, research on RPF has still much scope for progress, not only to remove the pressure on the species mentioned above, but also to find new candidate species for aquaculture by increasing efforts in the field of comparative reproductive biology of new species.
Finally, it may happen that advances in RPF knowledge open the door to innovative techniques that do not result of any demand from farmed fish producers and that can even be rejected by consumers in some countries, like transgenic fish for specific characteristics such as improved growth or disease resistance. In this field particularly, but also in the case of any innovation, public concern about biotechnological applications and sustainable aquaculture development must be taken into account.
Further Reading
| - | Access more information from Cybium by clicking here. |
October 2008

