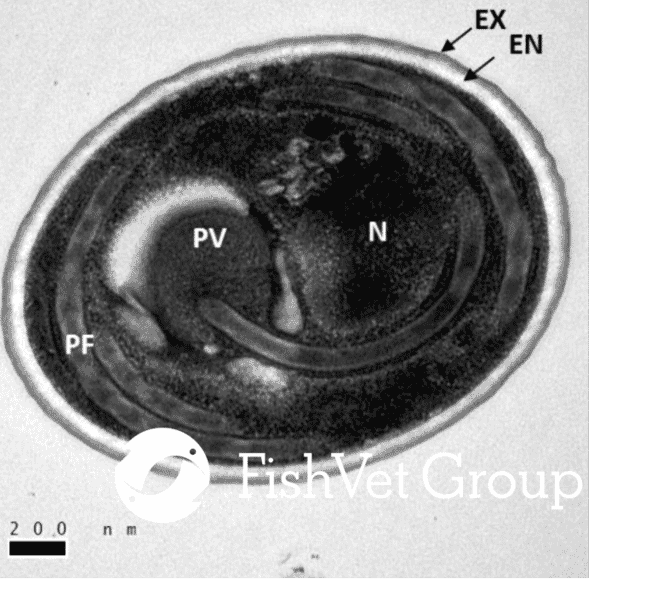
Microsporidia are spore-forming parasites, the exact classification of which has challenged scientists since their discovery in the 19th century. They are either directly responsible or are thought to play a role in many commercially significant diseases in aquaculture. A survey of 150 pre-stocked ponds in Thailand found prevalence of the microsporidian Enterocytozoon hepatopenaei at 49 percent, with the resultant economic losses due to slowed growth and early harvest estimated at $32,000 per hectare in every culture cycle.
Currently, there are 200 known genera of microsporidia, with over 80 known to infect fish and aquatic invertebrates. Although microsporidian infections have often been linked to immunocompromised individuals they are also well-known primary pathogens (ie declines in commercial fisheries have been attributed to microsporidiosis) and have been reported in immunocompetent patients, producing sub-clinical to lethal infections.
Salmonid infections
In aquaculture, several microsporidian species are known to cause economic losses to the industry. In Atlantic salmon, the microsporidian Desmozoon lepeophtherii may play a role in the multifactorial and often complex gill diseases which are increasingly being reported to affect fish in in Norway and Scotland. More recently, the parasite has been associated with growth retardation and work is underway to fully characterise the effect this pathogen has upon the production cycle. For salmonids cultured in marine net pens, different microsporidian species have been associated with mortalities: Loma salmonae have been shown to cause severe gill damage in Coho salmon (Oncorhynchus kisutch) and Chinook salmon (Oncorhynchus tshawytscha); Nucleopspora salmonis can infect hematopoietic cells, leading to a leukaemia-like condition among a broad range of salmonids; and Desmozoon lepeophtherii (syn. Paranucleospora theridion) has been suggested to be one of the pathogens involved in complex gill disease in Atlantic salmon (Salmo salar ).
Marine finfish
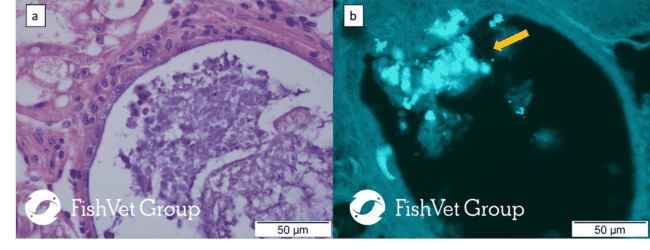
Cleanerfish species are also affected by these parasites, severe systemic infections and high mortalities have been recorded in the lumpsucker (Cyclopterus lumpus) due to Nucleospora cyclopteri. Other examples in finfish include the microsporidian Tetramicra brevifilum that infects the connective tissue of the skeletal musculature and causes significant disease in turbot (Scophtalmus maximus), reducing the growth rate in fish and leading to unmarketable fish due to skeletal muscle abnormalities, or Enterospora nucleophila in farmed sea bream (Sparus aurata) which causes significant mortalities and an emaciative syndrome. A significant example in microsporidians affecting farmed crustaceans is Enterocytozoon hepatopenaei (Figure 2), an emerging parasite and critical threat for farmed penaeid shrimp in Asia that causes severely retarded growth.
Diagnosis of microsporidians is mainly based on the use of molecular diagnosis methods and light microscopy to detect the spores. Microscopic visualization of microsporidians in tissue sections can be difficult due to the small size of the spores (~1-20µm). As a result, special staining methods such as modified trichrome stain, Gram, Warthin-Starry silver, or Calcofluor White may be required. More sensitive methods like immunohistochemistry or in situ hybridization are available for certain microsporidian species.
Different drugs have been tested to treat microsporidia infections, mostly on a research basis (ie benzimidazoles or fumagillin) but these have showed different results on different microsporidian species and hosts. The first successful anti-microsporidian vaccine was developed against Loma salmonae and demonstrated to reduce microsporidial gill disease, suggesting that a different approach could be followed for microsporidiosis disease control.